Search Results
Showing results 1 to 13 of 13

Separation Anxiety
Source Institutions
In this activity, learners discover the primary physical properties used to separate pure substances from mixtures.
Salts & Solubility
Source Institutions
In this online interactive simulation, learners will add different salts to water and then watch the salts dissolve and achieve a dynamic equilibrium with solid precipitate.

Lava Lamps
Source Institutions
Learners observe working lava lamps to understand how they work (included in PDF link).

To Dye For
Source Institutions
Learners add two dyes to mineral oil and water, and then compare their miscibility (how well they mix) in each.

Klutz-Proof Density Column
Source Institutions
Making liquids of different densities to perfectly lay on top of each other can be a frustrating exercise. The Exploratorium created this activity as a fool proof way of making a density column.
Foam Peanuts
Source Institutions
Learners compare the properties and solubilities of Styrofoam (TM), ecofoam packing peanuts, and popcorn. First, the solubility of each substance is tested in water.
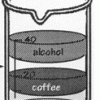
Layers of Liquids
Source Institutions
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.

The China Hammer Mystery
Source Institutions
In this activity, learners are asked to examine the differences between two materials in a pair.

Number of Mentos vs. Height: Soda Geyser Series #5
Source Institutions
In this activity, learners conduct a controlled experiment to examine how many Mentos are needed to make the tallest possible soda geyser.

Soda Brand vs. Height Experiment: Soda Geyser Series #4
Source Institutions
In this activity, learners conduct a controlled experiment to examine which brand of soda makes the best (highest) soda geyser.

What's the Matter
Source Institutions
In this activity, learners identify different classes of matter based on physical properties.

Swirling Milk
Source Institutions
In this chemistry activity, learners prepare two petri dishes, one filled with water and one filled with milk.

All Mixed Up!
Source Institutions
In this activity, learners separate a mixture of pebbles, salt crystals, and wood pieces. They add water and pour the mixture through a strainer.
