Search Results
Showing results 1 to 16 of 16

Funny Putty
Source Institutions
In this chemistry activity (page 1 of PDF SciGirls Activity: Milk Carton Boat), learners will create a blob of stretchable funny putty out of a water, borax, and glue mixture.
Monster Mallows
Source Institutions
In this activity, learners explore how ordinary marshmallows expand when heated in a microwave.
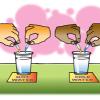
Molecules in Motion
Source Institutions
In this activity, learners add food coloring to hot and cold water to see whether heating or cooling affects the speed of water molecules.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

Soapy Boat
Source Institutions
Learners discover that soap can be used to power a boat. Learners make a simple, flat boat model, put it in water, and then add a drop of detergent at the back of the boat.

Kool Colors
Source Institutions
Learn about dyes and mordants (fixatives) when you tie-dye fabric with Kool-Aid™ and vinegar. The colored molecules in Kool-Aid™ form a chemical bond between the fiber and dye molecules.
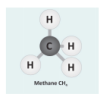
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Toast a Mole!
Source Institutions
In this quick activity, learners drink Avogadro's number worth of molecules - 6.02x10^23 molecules!

Copper Caper
Source Institutions
In this activity, learners conduct an oxidation experiment that turns old pennies bright and shiny. Learners soak 20 dull, dirty pennies in a bowl of salt and vinegar for five minutes.

The Ups and Downs of Thermometers
Source Institutions
In this activity, learners examine the parts of a thermometer. After placing a thermometer in hot and cold water, learners look at molecular model animations of the liquid in a thermometer.

Battling for Oxygen
Working in groups, learners model the continuous destruction and creation of ozone (O3) molecules, which occur in the ozone layer.

Stained Glass Glue
Source Institutions
In this activity on page 6 of the PDF, learners use glue instead of glass to create artwork that can be hung in a window.
3D-ection: Molecular Shape Recognition
Source Institutions
In this activity (page 12), learners explore how molecules self-assemble and how molecules must fit together, like a lock and key, in order to identify each other and initiate a new function as a comb

A Slime By Any Other Name
Source Institutions
This fun video explains how to make a batch of oobleck (or slime) and why this special substance is known as a "non-Newtonian" fluid. Watch as Mr.

Moving Molecules!
Source Institutions
In this activity about molecular diffusion (located on page 2 of the PDF under Nanosilver Activity), learners will make predictions and move molecules of iodine through a seemingly solid plastic sandw

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.
