Search Results
Showing results 1 to 20 of 28

Hot Equator, Cold Poles
Source Institutions
In this activity, learners use multiple thermometers, placed at different angles, and a lamp to investigate why some places on Earth's surface are much hotter than others.
Making a One-Second Timer
Source Institutions
This lab activity has learners create a pendulum with a one-second period.
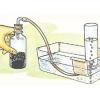
Pneumatic Trough
Source Institutions
In this activity, learners build a "pneumatic trough," a laboratory apparatus used for collecting pure gas samples over water.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Counting With Quadrants
Source Institutions
Millions of organisms can live in and around a body of water.

Wild Sourdough
Source Institutions
In this activity, learners explore chemistry and the microbial world by making their own sourdough starter and bread at home using only flour and water.

Forgotten Genius
Source Institutions
This series of chemistry stations is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Each of the six stations features either a chemical or p
Transformation of E. coli Using Green Fluorescent Protein
Source Institutions
In this activity related to plant biotechnology, learners transform a strain of E. coli using green fluorescent protein from a bioluminescent jellies.

Does Sunscreen Protect My DNA?
Source Institutions
In this laboratory experiment, learners explore how effectively different sunscreens protect yeast cells from damage caused by ultraviolet (UV) radiation.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Edible Glass
Source Institutions
In this activity, learners discover the principles of edible glass by making a supersaturated sugar solution.

Seas in Motion
Source Institutions
In this outdoor, beach activity, learners use tennis balls, water balloons and other simple devices to investigate the movement of waves and currents off a sandy beach.

Potato Power
Source Institutions
Learners combine hydrogen peroxide with three different forms of potato: raw chunks, ground chunks, and boiled chunks.

Think Fast!: Just How Quick Are You?
Source Institutions
This is an activity about reaction times. Just how quickly must an NHL goalie respond to save a shot, and how does your reaction time compare?
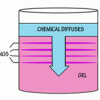
Liesegang Rings
Source Institutions
This display shows slow chemical reactions in colorful crystal formations known as Liesegang Rings. These reactions are similar to those forming the rings in agates.
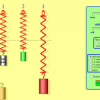
Masses & Springs
Source Institutions
In this online activity, learners use a realistic mass and spring laboratory. They hang masses from springs and adjust the spring stiffness and damping.

Invisible Sunblock
Source Institutions
This is a hands-on activity exploring how nanoscale particles are used in mineral sunblocks to increase their transparency.

Demonstrating An Epidemic
Source Institutions
This experiment allows learners to experience a small scale "epidemic," demonstrating the ease with which disease organisms are spread, and enables learners to determine the originator of the "epidemi
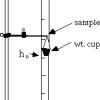
Stretching Wires
Source Institutions
In this activity, learners determine the elastic and plastic properties of different types of metal wires.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.
