Search Results
Showing results 1 to 20 of 26
Trading Places: Redox Reactions
Source Institutions
Visitors add drops of copper sulfate solution onto a steel nail. They observe the nail change color from silver to brown as the copper plates onto the nail.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

Setting the Scene
Source Institutions
In this activity (on page 2), pairs of learners create an imaginary crime scene. One person leaves the room while the other person moves a few things around.

Fingerprint Identification
Source Institutions
In this activity (on page 2) about fingerprint analysis, learners use graphite from a pencil and scotch tape to capture their fingerprints.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
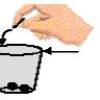
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Density Rainbow
Source Institutions
In this activity, learners mix several sugar solutions to investigate the property of density. Each sugar solution has a different density and color of the rainbow.
Building a Community From the Ground Up
Source Institutions
In this activity, a group of learners work collaboratively to design and construct a paper model showing the evolution of an environment through multiple stages, from prehistory through the modern cit
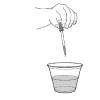
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d
Concentrate: Concentrations and Reaction Rates
Source Institutions
Visitors incrementally increase the amount of iodate in three different test tubes containing the same amount of a starch solution.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.
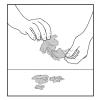
Busted by Biology
Source Institutions
In this two-part activity, learners will extract their own DNA from their cheek cells and learn how DNA is analyzed and used to solve crimes.

Forwards and Backwards
Source Institutions
In this activity, learners explore acids and bases by preparing six solutions that combine vinegar and ammonia, ranging from acid (all vinegar) to base (all ammonia).

Three Little Pigs
Source Institutions
In this activity, leaners explore building techniques by recreating the story of The Three Little Pigs.
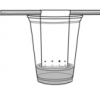
Water Ways
Source Institutions
In this activity (on page 2 of the PDF), learners explore surface tension by adding pennies to cups which are "full" of plain water or soapy water.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.
