Search Results
Showing results 1 to 11 of 11
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.

Water Body Salinities I
Source Institutions
In this activity, learners investigate the different salinity levels of oceans, rivers and estuaries.

Conductivity Meter
Source Institutions
In this activity, learners build a simple qualitative conductivity tester with a battery, bulb and foil.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.

Forms of Carbon
Source Institutions
In this activity, educators can demonstrate how the nanoscale arrangement of atoms dramatically impacts a material’s macroscale behavior.

Make Your Own Batteries!
Source Institutions
This activity (on page 3 of the PDF under GPS: Body Electricity Activity) is a full inquiry investigation into conductivity.
Properties of Metals
Source Institutions
In this activity, learners explore the properties of metals at four stations. The stations include A) Magnetism and Breakfast Cereal; B) Conductivity of Metals; C) Alloys; and D) Metal Plating.

Carrying Charges: Testing for Conductivity
Source Institutions
Learners are challenged to create solutions that conduct electricity and make a buzzer buzz (or an LED light up).

Balloon Nanotubes Tabletop
Source Institutions
This activity introduces learners to the structure and properties of carbon nanotubes.
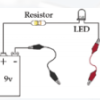
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.

The Power of Graphene
Source Institutions
This lesson focuses on graphene and its electrical properties and applications.
