Search Results
Showing results 1 to 7 of 7

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.
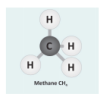
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Toast a Mole!
Source Institutions
In this quick activity, learners drink Avogadro's number worth of molecules - 6.02x10^23 molecules!
What's So Special about Water: Surface Tension
Source Institutions
In this three-part activity, learners play a game and conduct two simple experiments to explore water and surface tension. Learners will have fun discovering how water "sticks" together.

Battling for Oxygen
Working in groups, learners model the continuous destruction and creation of ozone (O3) molecules, which occur in the ozone layer.

Water Cycle in a Bag
Source Institutions
In this activity, learners create a biosphere in a baggie.

Pepper Scatter
Source Institutions
In this quick activity, learners break the tension that happens when water develops a "skin." Learners use water, pepper and some soap to discover the wonders of surface tension—the force that attract
