Search Results
Showing results 1 to 8 of 8

Electroplating
Source Institutions
In this electrochemistry activity, learners will explore two examples of electroplating.

Ripening of Fruits and Vegetables
Source Institutions
In this activity, learners test the rate of ripening fruit and vegetables and use a chemical to inhibit the ripening process.

Fuel for Living Things
Source Institutions
In this activity, learners observe what happens when yeast cells are provided with a source of food (sugar). Red cabbage "juice" will serve as an indicator for the presence of carbon dioxide.

Can You Copperplate?
Source Institutions
In this activity, learners explore chemical engineering and how the processes of chemical plating and electroplating have impacted many industries.

Copper Caper
Source Institutions
In this activity, learners conduct an oxidation experiment that turns old pennies bright and shiny. Learners soak 20 dull, dirty pennies in a bowl of salt and vinegar for five minutes.
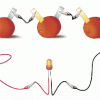
Electricity: Fruit Batteries
Source Institutions
In this activity, learners create a battery from fruit. This activity helps learners explore electricity, electrochemistry, and series circuits as well as the process of scientific inquiry.

Making Sodium Acetate: Hot Ice
Source Institutions
In this chemistry activity which should only be done under adult supervision (page 10 of the PDF), learners will create an exothermic process by making Sodium Acetate.

Off Base
Source Institutions
In this activity, learners explore the factors that tend to resist changes in pH of the ocean and why the ocean is becoming more acidic.
