Search Results
Showing results 1 to 20 of 53

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Newspaper Collage
Source Institutions
In this activity on page 3 of the PDF, learners create a collage by using vinegar to transfer color pictures from a newspaper onto a piece of white paper.

Art with Salt and Ice
Source Institutions
This open-ended art project allows learners to create their own colorful ice sculpture by using rock salt and food coloring on a solid block of ice.

How Can Gravity Make Something Go Up?
Source Institutions
In this activity, learners use cheap, thin plastic garbage bags to quickly build a solar hot air balloon. In doing so, learners will explore why hot air rises.

Descartes' Diver
Source Institutions
In this activity, learners explore how changes in fluid pressure affect the buoyancy of a Cartesian diver inside a plastic soda bottle.

Exploring How Liquids Behave
Source Institutions
Learners apply their knowledge from a previous study to identify different liquids--water, corn syrup, and vegetable oil.
Exploring Liquids
Source Institutions
Young learners investigate and observe the properties of three liquids -- water, vegetable oil, and corn syrup. They use their senses to collect data and ask and answer questions.

Polymers are Chains (K-2)
Source Institutions
In this activity, learners make a paper model of a polymer, then make Silly Putty, an actual polymer.

Floating Dry Erase Creations
Source Institutions
In this activity, learners will create a drawing with dry erase markers and watch it come to life. Learners will explore chemistry, art and storytelling through this activity.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.
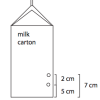
Pressing Pressure
Source Institutions
In this activity, learners compare water pressure at different depths. Learners discover that water pressure increases with depth.

What Counts in Bounce
Source Institutions
In this activity learners compare the bounciness of warm and cold racquetballs to see if temperature makes a difference in how well they bounce.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.
Getting Windy
Source Institutions
In this activity, learners create and understand surface currents. Learners create example surface currents and discover how landmasses affect the current.

A Dissolving Challenge
Source Institutions
In this activity, learners add objects and substances to carbonated water to discover that added objects increase the rate at which dissolved gas comes out of solution.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.

Ziptop Bag Chemistry
Source Institutions
In this chemistry activity, learners perform three chemical reactions in a sealed zip-top bag. Learners will record their observations and classify the changes as chemical or physical.

New Sense about Cents
Source Institutions
In this activity on page 6 of the PDF (Chemistry—It’s Elemental), learners explore some of the properties of copper using a few common household ingredients.

Moving Molecules!
Source Institutions
In this activity about molecular diffusion (located on page 2 of the PDF under Nanosilver Activity), learners will make predictions and move molecules of iodine through a seemingly solid plastic sandw
