Search Results
Showing results 161 to 180 of 206

Wet Pennies
Source Institutions
Learners initially test to see how many drops of liquid (water, rubbing alcohol, and vegetable oil) can fit on a penny.

Pea Brain!: Explorations in Estimation
Source Institutions
In this activity, learners use two different techniques to estimate how many little things fit into one bigger thing.

Paper Lanterns
Source Institutions
In this activity, learners explore light and shadows by creating a lantern they can keep on their nightstand.
M&M® Model of the Atom: Edible Subatomic Particles
Source Institutions
In this activity, learners use colored candy to represent subatomic particles and make a model of an atom (Bohr model).

Cipher Wheel
Source Institutions
In this activity, learners make their own encrypted code to pass along secret messages using a printable cipher wheel.

Sweet Measurements
Source Institutions
In this activity on page 3 of the PDF, learners investigate how much sugar is in a soda. Learners use sugar cubes to measure and calculate the amount of sugar in a bottle of soda.

Inflate-a-mole
Source Institutions
In this activity, learners conduct an experiment to find the volume of one mole of gas. Learners capture sublimated gas from dry ice in a ziploc bag and use water displacement to measure its volume.

Polarized Sunglasses
Source Institutions
In this activity, learners explore how polarizing sunglasses can help diminish road glare.

Does Your Chewing Gum Lose Its Flavor?
Source Institutions
Each learner chews a piece of gum until it loses its flavor, and then leaves the gum to dry for several days.

If Anyone Can, Icon
Source Institutions
In this activity, learners create their own icons for a forecast-at-a-glance poster for their classroom/learning space.

Where on Earth?
Source Institutions
Learners look at labels on items in the room to determine where each item was manufactured, then write about the item on an index card.

Double Replacement Micro-Reactions
Source Institutions
In this chemistry activity, learners use common chemicals to examine reactions that occur between two aqueous solutions.

Algebra: Aw Chute!
Source Institutions
In this math lesson, learners determine and compare the rate of descent of various learner-constructed parachutes. Learners construct parachutes that will have maximum hang times.

Solar System on a Stick
Source Institutions
Learners build a model of the planets in the solar system. In their model, the planets are spaced in their relative distance from the Sun.
Alien Encounter
Source Institutions
Use multiplication to help the aliens find out how much fuel they need to get their spacecrafts home.

What am I?
Source Institutions
In this activity, learners examine nanoscale structures of common things.

Up, Up, and Away
Source Institutions
In this math lesson, learners explore the real world meaning of slope. Learners conduct a balloon experiment in which they measure the circumference and flight time of deflating balloons.
Cryptographic Protocols: The Peruvian Coin Flip
Source Institutions
This activity about cryptographic techniques illustrates how to accomplish a simple, but nevertheless seemingly impossible task—making a fair, random choice by flipping a coin between two people who d

Unlocking the Secret in Product Codes
Source Institutions
In this activity, learners will explore how product barcodes are scanned accurately and be able to "guess" the last number in a 13 digit barcode correctly every time.
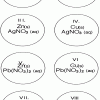
Single Replacement Micro-Reactions
Source Institutions
In this chemistry activity, learners use common chemicals and metals to explore single replacement reactions.
