Search Results
Showing results 21 to 40 of 53

Rock Bottoms
Source Institutions
Learners add acid rain (nitric acid) to two cups that represent lakes. One cup contains limestone gravel and the other contains granite gravel.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.
Plastic Milk: You can make plastic from milk
Source Institutions
In this activity (on page 2 of the PDF), learners make a plastic protein polymer from milk. Adding vinegar to milk causes the protein casein to solidify or curdle.

Draggin' Boats
Source Institutions
Learners design, build, and test models of "dragon boats" made from up to three milk cartons.

Big Things Come in Little Packages
Source Institutions
As a group, learners investigate three packages which are all the same size and shape, but have different contents. One is filled with foam, one is filled with wood, and one is filled with metal.
Layers of Liquids
Source Institutions
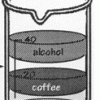
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.

Oh Buoy!
Source Institutions
Learners work in pairs to design, construct, and test a device that exhibits positive, neutral, and negative buoyancy. They test a number of different objects in water to see if they sink or float.

Burning Issues
Source Institutions
Learners use a candle to investigate the products of combustion. When a glass rod is held over a lit candle, the candle flame deposits carbon on the rod.

Curious Contraptions
Source Institutions
In this engineering design activity, learners will design, test, and build a “haunting machine” to solve a Sherlockian mystery.
Busted by Biology
Source Institutions
In this two-part activity, learners will extract their own DNA from their cheek cells and learn how DNA is analyzed and used to solve crimes.

A Matter of Splatter
Source Institutions
In this math-based activity, learners will experiment to find how height and angle affect spatter and then use this knowledge to solve a crime.
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

What's Your Blood Type?
Source Institutions
In this activity, learners perform a simulated blood test procedure.
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d

Trading Places
Source Institutions
In this activity, learners discover that atoms and ions of different metals will change places.
Floating Golf Ball
Source Institutions
Visitors observe a graduated cylinder with a golf ball floating about halfway in liquid. The bottom half of the cylinder contains a concentrated solution of salt.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.

Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.
