Search Results
Showing results 1 to 20 of 64

Fizzy Nano Challenge
Source Institutions
This lesson focuses on how materials behave differently as their surface area increases.

Tiny Tubes
Source Institutions
In this activity, learners make "totally tubular" forms of carbon. Learners use chicken wire to build macro models of carbon nanotubes.

Surface Tension Icebreaker
Source Institutions
This is a quick activity (located on page 2 of the PDF under Nasturtium Leaves Activity) about surface tension.
Water Clean-up
Source Institutions
This is an activity (located on page 3 of the PDF under Water Clean-up Activity) about the use of reduction agents to decontaminate ground water.

Surface Area and Soda Geysers
Source Institutions
This is an activity (located on page 4 of the PDF under Surface Area Activity) about surface area and reactivity.

Jell-O Model of Microfluidics
Source Institutions
This activity uses Jell-O(R) to introduce learners to microfluidics, the flow of fluids through microscopic channels.

Be a Scanning Probe Microscope
Source Institutions
In this activity, learners investigate Scanning Probe Microscopes (SPM) and then work in teams using a pencil to explore and identify the shape of objects they cannot see, just as SPMs do at the nano

Morphing Butterfly
Source Institutions
In this activity, learners explore how nanosized structures can create brilliant color.

What is Nanotechnology?
Source Institutions
In this activity related to nanotechnology, learners observe some of the effects that result from creating a thin layer of material several nanometers thick.

Composite Materials
Source Institutions
This is an activity (located on page 3 of PDF under Hockey Sticks Activity) about composites, materials made of 2 or more different components.

Gravity Fail
Source Institutions
In this activity, learners try pouring water out of a regular cup and a miniature cup. It’s harder than it sounds! Learners discover that different forces dominate at different size scales.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Iridescent Art
Source Institutions
This is a quick activity (on page 2 of the PDF under Butterfly Wings Activity) that illustrates how nanoscale structures, so small they're practically invisible, can produce visible/colorful effects.

How Big is Small
Source Institutions
In this classic hands-on activity, learners estimate the length of a molecule by floating a fatty acid (oleic acid) on water.
Beam Me Up!
Source Institutions
This is a quick activity (on page 2 of the PDF under Stained Glass Activity) about the "Tyndall effect," the scattering of visible light when it hits very small dispersed particles.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Invisible Sunblock
Source Institutions
In this activity, learners find out why some mineral sunblock rubs in clear. Learners compare nano and non-nano sunblocks and discover how particle size affects visibility.
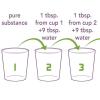
Sniffing for a Billionth
Source Institutions
This is an activity (located on page 4 of the PDF under What's Nano? Activity) about size and scale.

Exploring at the Nanoscale
Source Institutions
This lesson focuses on how nanotechnology has impacted our society and how engineers have learned to explore the world at the nanoscale.

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.
