Search Results
Showing results 1 to 13 of 13

Spectroscope
Source Institutions
In this activity (posted on March 12, 2011), learners follow the steps to construct a spectroscope, a tool used to analyze light and color.
First Impressions
Source Institutions
Learners experiment with a commercial photo-sensitive paper (Sunprint® or NaturePrint® paper). They place opaque and clear objects on the paper and expose it to bright light, observing the results.
Beam Me Up!
Source Institutions
This is a quick activity (on page 2 of the PDF under Stained Glass Activity) about the "Tyndall effect," the scattering of visible light when it hits very small dispersed particles.

Pearlescent Pigments
Source Institutions
This is written as a display, but can easily be adapted to a hands-on activity. Learners observe and shake containers of shiny liquids.

Photolithography
Source Institutions
In this activity, learners use UV light to transfer a pattern onto a plastic board. The pattern is transferred by placing a mask (a transparency sheet with the pattern) on a plastic board.

Glow Up
Source Institutions
In this activity, learners explore chemiluminescence and fluorescence. Learners examine 3 different solutions in regular light, in the dark with added bleach solution, and under a black light.

Battling for Oxygen
Working in groups, learners model the continuous destruction and creation of ozone (O3) molecules, which occur in the ozone layer.

Fruity Electricity
Source Institutions
In this activity, Frankenstein's lab is running out of electricity! Learners use fruit to help Igor find a temporary source of energy to turn on a light.

Exploring Materials: Graphene
Source Institutions
In this activity, learners investigate the properties of graphene and graphite.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.

Penny Battery
Source Institutions
In this activity, learners light an LED with five cents. Learners use two different metals and some sour, salty water to create a cheap battery.

Science of Sunblock
Source Institutions
This is an activity (located on page 3 of the PDF under Stained Glass Activity) about nanotechnology making its way into everyday products, such as sunscreen, and how effective these invisible particl
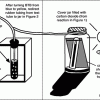
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.
