Search Results
Showing results 1 to 20 of 281
Water Ways
Source Institutions
In this activity (on page 2 of the PDF), learners explore surface tension by adding pennies to cups which are "full" of plain water or soapy water.
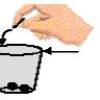
When is a Glass of Water Really Full?
Source Institutions
In this activity, learners see how many coins they can add to a full glass of water before the water overflows.

Clean Water: Is It Drinkable?
Source Institutions
In this activity, learners simulate nature's water filtration system by devising a system that will filter out both visible and invisible pollutants from water.

Cleaning Water with Dirt
Source Institutions
In this activity on page 7 of the PDF (Water in Our World), learners make their own water treatment systems for cleaning water.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

"Boyle-ing" Water
Source Institutions
In this activity, learners explore Boyle's Law and discover that water will boil at room temperature if its pressure is lowered.

Breaking the Tension: Surface Tension 1
Source Institutions
Learners explore how the attractive forces between water molecules create surface tension and allow certain objects to float on the surface of water.

Drop Shape
Source Institutions
In this activity, learners get a closer look at the shape of a drop of water and a drop of oil. Learners first drip water onto wax paper and examine the shape of separate drops from a side view.

Comparing the Density of an Object to the Density of Water
Source Institutions
Learners compare the weight of equal volumes of wax, water, and clay. Learners discover that since the wax weighs less than an equal volume of water, it is less dense than water and will float.

Water Treatment
Source Institutions
Water treatment on a large scale enables the supply of clean drinking water to communities.

Diffusion of Water with Gummy Bears
Source Institutions
In this activity, learners investigate the movement of water into and out of a polymer. Learners test the diffusion of water through gummy bears, which are made of sugar and gelatin (a polymer).

Penny Drop
Source Institutions
In this quick activity about the properties of water (page 1 of PDF under SciGirls Activity: Malformed Frogs), learners will use an eyedropper to slowly place one drop of water at a time onto a penny,

Changing the Density of a Liquid: Heating and Cooling
Source Institutions
Learners investigate how the temperature of water affects its density.

Disappearing Crystals
Source Institutions
Learners experiment with water gel crystals, or sodium polyacrylate crystals, which absorb hundreds of times their weight in water. When in pure water, the water gel crystals cannot be seen.

Moving On Up: Capillary Action 1
Source Institutions
Over the course of several days, learners explore the property of water that helps plants move water from roots to leaves or gives paper towels the capacity to soak up water.

Electrostatic Water Attraction
Source Institutions
In this activity, learners conduct a simple experiment to see how electrically charged things like plastic attract electrically neutral things like water.

Stuck on You: Adhesion
Source Institutions
Learners explore water adhesion and learn about why water molecules are more strongly attracted to some substances than others.

Sunny Day Painting
Source Institutions
In this activity, learners explore properties of water and watch evaporation happen by "painting" with water in the sun.
Water Holes to Mini-Ponds
Source Institutions
Dig a hole, line it, fill it with fresh water, and you have a water hole: a good place to study colonization.
What's So Special about Water: Absorption
Source Institutions
In this activity about water's cohesive and adhesive properties and why water molecules are attracted to each other, learners test if objects repel or absorb water.
