Search Results
Showing results 1 to 20 of 49

Acid (and Base) Rainbows
Learners use red cabbage juice and pH indicator paper to test the acidity and basicity of household materials. The activity links this concept of acids and bases to acid rain and other pollutants.

Modeling Day and Night
Source Institutions
In this activity (on page 1 of the PDF), learners make a "mini-globe" to investigate the causes of day and night on our planet.

Straining Out the Dirt
Learners take on the role of environmental engineers as they design water filters.

Fragile Waters
Source Institutions
In this activity (on pages 18-29) learners explore the impact of the March 24, 1989 oil spill in Alaska caused by the Exxon Valdez tanker.
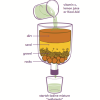
Water Clean-up
Source Institutions
This is an activity (located on page 3 of the PDF under Water Clean-up Activity) about the use of reduction agents to decontaminate ground water.

What's Hiding in the Air?: Acid Rain Activity
As a model of acid rain, learners water plants with three different solutions: water only, vinegar only, vinegar-water mixture.

What's In Your Breath?
Source Institutions
In this activity, learners test to see if carbon dioxide is present in the air we breathe in and out by using a detector made from red cabbage.

Capturing Homemade Microgravity
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Microgravity) is a full inquiry investigation into how ordinary things behave in microgravity, similar to what astronauts experience.

My Angle on Cooling: Effects of Distance and Inclination
Source Institutions
In this activity, learners discover that one way to cool an object in the presence of a heat source is to increase the distance from it or change the angle at which it is faced.

Crystal Gardens
Source Institutions
In this activity, which requires adult supervision, learners get to explore the awesome power of chemistry.

Water Quality and pH Levels in Aquatic Ecosystems
Source Institutions
In this fun and in depth hands-on experiment, learners test various liquid samples (distilled water, lemon juice, vinegar, and baking soda mixed with water) to determine their pH levels and identify e

Corals and Chemistry
Source Institutions
In this activity, learners investigate how increased carbon dioxide (CO2) emissions from the burning of fossil fuels is changing the acidity (pH) of the ocean and affecting coral reefs and other marin

What does Color have to do with Cooling?
Source Institutions
In this demonstration/experiment, learners discover that different colors and materials (metals, fabrics, paints) radiate different amounts of energy and therefore, cool at different rates.
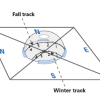
Reason for the Seasons
Source Institutions
In this activity (on page 6 of the PDF), learners plot the path of the sun's apparent movement across the sky on two days, with the second day occurring two or three months after the first.
Coral and Chemistry
Source Institutions
In this experiment, learners will explore whether increased carbon dioxide makes our oceans more basic or more acidic.
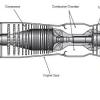
Jet Propulsion
Source Institutions
In this two-part activity, learners work in pairs to examine the four basic stages of a turbine engine.

Salt Water Revival
Source Institutions
In this outdoor activity, learners visit the intertidal zone of a rocky coastal site well populated with marine organisms.

Burning Issues
Source Institutions
Learners use a candle to investigate the products of combustion. When a glass rod is held over a lit candle, the candle flame deposits carbon on the rod.

Oil Spill Solutions
Source Institutions
In this activity, learners explore how environmental engineers might approach solving the problem of an oil spill.

What is in the Water?
Source Institutions
In this activity, learners use open inquiry to learn about the process of science as well as gain experience regarding the Law of Conservation of Mass, dissolution, and density.
