Search Results
Showing results 1 to 20 of 122

Tiny Tubes
Source Institutions
In this activity, learners make "totally tubular" forms of carbon. Learners use chicken wire to build macro models of carbon nanotubes.

Newspaper Collage
Source Institutions
In this activity on page 3 of the PDF, learners create a collage by using vinegar to transfer color pictures from a newspaper onto a piece of white paper.

Exploring Products: Nano Sand
Source Institutions
In this activity, learners explore how water behaves differently when it comes in contact with "nano sand" and regular sand.

Exploring Materials: Liquid Crystals
Source Institutions
In this activity, learners discover that the way a material behaves on the macroscale is affected by its structure on the nanoscale.

Liquid Body Armor
Source Institutions
In this activity, learners explore how nanotechnology is being used to create new types of protective fabrics.

Watching Crystals Grow
Source Institutions
Learners will compare the growth rate and appearance of crystals forming on small rocks to those growing on miscellaneous objects. Learners will also investigate how temperature (warm vs.

Surface Tension Icebreaker
Source Institutions
This is a quick activity (located on page 2 of the PDF under Nasturtium Leaves Activity) about surface tension.
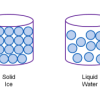
Atoms and Matter (3-6)
Source Institutions
In this activity, learners build models of atoms and molecules, then consider their role in different phases of matter, density, and mixtures and solutions.

Clear Slime Polymer
Source Institutions
In this chemistry activity, learners use guar gum to make slime. Use this activity to introduce learners to polymers, viscosity, and colloids.

Funny Putty
Source Institutions
In this chemistry activity (page 1 of PDF SciGirls Activity: Milk Carton Boat), learners will create a blob of stretchable funny putty out of a water, borax, and glue mixture.

Gravity Fail
Source Institutions
In this activity, learners try pouring water out of a regular cup and a miniature cup. It’s harder than it sounds! Learners discover that different forces dominate at different size scales.

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.

Orange Pop
Source Institutions
In this activity, learners surprisingly pop balloons without touching them using orange peels. This Mr.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

The Carbon Cycle and its Role in Climate Change: Activity 3
Source Institutions
In this activity, learners explore the human influences on the carbon cycle and examine how fossil fuels release carbon.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.
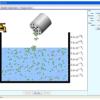
Salts & Solubility
Source Institutions
In this online interactive simulation, learners will add different salts to water and then watch the salts dissolve and achieve a dynamic equilibrium with solid precipitate.

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.

Bubble Trouble
Source Institutions
In this activity on page 15 of the PDF, learners measure the amount of bubbles that they make using a detergent.

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).
