Search Results
Showing results 1 to 14 of 14

Illuminations on Rates of Reactions
Source Institutions
In this activity, learners investigate the speed of chemical reactions with light sticks. Learners discover that reactions can be sped up or slowed down due to temperature changes.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Lava Lamps
Source Institutions
Learners observe working lava lamps to understand how they work (included in PDF link).

Plaster of Paris
Source Institutions
In this activity (page 6 of the PDF), learners will observe both a chemical and a physical change.

Pie-Pan Convection
Source Institutions
It's difficult to see convection currents in any liquid that's undergoing a temperature change, but in this Exploratorium Science Snack, you can see the currents with the help of food coloring.

Temperature vs. Height: Soda Geyser Series #6
Source Institutions
In this activity, learners conduct a controlled experiment to examine how temperature will affect the height of a soda geyser.

Instant Ice Cream with a Dry Ice Bath
Source Institutions
In this chemistry meets cooking activity, learners make carbonated, vanilla ice cream using dry ice and denatured ethanol, which are both inexpensive and accessible.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
Sodium Acetate Hand Warmers
Source Institutions
In this activity, sodium acetate hand warmers are used to introduce learners to supersaturated solutions, crystallization, and exothermic reactions.
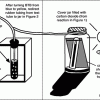
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Geyser
Source Institutions
This Exploratorium activity can be used in many contexts because geysers are great opportunities for learning about heat and temperature changes as well as geological/space science phenomena.

What's in the Water
Source Institutions
"What's in the Water" lets participants use tools to solve the mystery- what chemicals and compounds are in a sample of water?

Keep it Cool
Source Institutions
In this activity, learners explore how engineers have met the challenge of keeping foods, liquids, and other items cool.
