Search Results
Showing results 1 to 20 of 42

Rotating Light
Source Institutions
In this activity, learners explore what happens when polarized white light passes through a sugar solution.

Luminol Test
Source Institutions
Learners mix a solution containing luminol and copper with a fake blood solution. A chemical reaction between the luminol solution and fake blood (hydrogen peroxide) show learners a blue glow.
Pencil Drop
Source Institutions
In this demonstration, learners observe as a bottle is placed on a table with wooden hoop balanced on top and a pencil balanced on top of the hoop.

Amazing Marshmallows
Source Institutions
In this demonstration, learners observe the effects of air pressure. They will watch as marshmallows inside a bottle expand as a vacuum pump removes air from the bottle.

Eddy Currents
Source Institutions
In this activity related to magnetism and electricity, learners discover that a magnet falls more slowly through a metallic tube than it does through a nonmetallic tube.

Rubber Band Thermodynamics
Source Institutions
In this demonstration, learners explore the thermal properties of rubber. Learners investigate whether a rubber band contracts or expands when heated.

Egg Drop
Source Institutions
Perform this classic inertia demonstration to illustrate the transfer of potential energy to kinetic energy.

Do Cities Affect the Weather?
Source Institutions
In this activity, learners explore clouds and how they form.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

As The Stomach Churns
Source Institutions
In this chemistry activity, learners fill two test tubes with a solution of "artificial stomach fluid," consisting of hydrochloric acid in the same concentration as in human stomachs, some soap to cre

Natural Buffers
Source Institutions
Learners use a universal indicator to test the amount of sodium hydroxide needed to change the pH of plain water compared with the amount needed to change the pH of gelatin.

Common Scents
Source Institutions
Learners use a mortar and pestle to extract clove oil from cloves using denatured alcohol. They put this oil on paper, which they can take home.
Pepper Scatter
Source Institutions
In this activity, learners explore the forces at work in water. Learners experiment to find out what happens to pepper in water when they touch it with bar soap and liquid detergent.

Red, White and Blue I Demonstration
Source Institutions
In this chemistry demonstration, learners observe a chemical reaction that produces a colorful effect.

Transit Tracks
Source Institutions
In this space science activity, learners explore transits and the conditions when a transit may be seen.
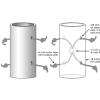
Mystery Tubes
Source Institutions
Learners investigate a pre-constructed mystery tube to determine its interior mechanism.

Concentrate!
Source Institutions
In this investigation of reaction kinetics, learners alter the amount of iodate solution mixed with the same amount of starch solution.

A Mole of Gas
Source Institutions
In this two-part activity, learners use everyday materials to visualize one mole of gas or 22.4 liters of gas. The first activity involves sublimating dry ice in large garbage bag.

Inverted Foucault Pendulum
Source Institutions
In this demonstration, learners explore a variation of a Foucault pendulum, but upside down.

Acid Rain
Source Institutions
In this chemistry demonstration, acid rain is simulated in a petri dish.
