Search Results
Showing results 21 to 34 of 34
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Model Wind Tunnel
Source Institutions
In this activity, learners build a miniature wind tunnel to measure force. Learners construct the model out of Lexan plastic, a fan, and a precise digital scale.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Energy Choices Board Game
Source Institutions
This board game teaches learners about energy decision making. Players select cards that determine the transportation and home design that will influence their expenses as they play.
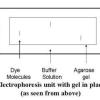
Gel Electrophoresis of Dyes
Source Institutions
In this experiment related to plant biotechnology, learners discover how to prepare and load an electrophoresis gel.
Shrinkers
Source Institutions
Visitors use heat to shrink samples of polystyrene. They compare samples from containers that were shaped in different ways during manufacturing.

Laser Projection Microscope
Source Institutions
In this activity, learners use a laser pointer to project a microscopic image of a liquid sample suspended from the tip of a syringe.
Concentrate: Concentrations and Reaction Rates
Source Institutions
Visitors incrementally increase the amount of iodate in three different test tubes containing the same amount of a starch solution.
Great Steamboat Race
Source Institutions
In this outdoor activity, learners race small boats, made of cork, balsa wood, popsicle sticks etc., to investigate the rate and direction of currents in a stream or creek.

Magic Inks
Source Institutions
Learners write their initials by applying different clear "magic ink" solutions to separate pieces of paper and then "develop" the inks with other clear solutions.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.

Flubber
Source Institutions
Learners experiment with a piece of Silly Putty® by stretching, bouncing, and snapping it. They then create flubber, a similar substance, by mixing diluted glue and a solution of sodium borate.

Electroplating
Source Institutions
In this activity, learners electrically plate zinc onto brass objects.
