Search Results
Showing results 1 to 20 of 28

Metal Reactions
Source Institutions
This is written as a static display, but can easily be adapted to a hands-on experiment for learners to conduct.

Hot Equator, Cold Poles
Source Institutions
In this activity, learners use multiple thermometers, placed at different angles, and a lamp to investigate why some places on Earth's surface are much hotter than others.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.

Fireworks!
Source Institutions
In this chemistry lab activity, learners model the colors of fireworks by burning metallic solutions in a flame and observing the different colors produced.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Rocket Reactions
Source Institutions
The "Rocket Reactions" activity is an exciting way to learn about how materials interact, behave, and change.

Power To Go
Source Institutions
Learners observe an electrochemical cell constructed from a small jar containing zinc and copper strips immersed in separate solutions. The strips are connected to a motor that turns a small fan.
Egg Osmosis
Source Institutions
Visitors observe three beakers. One beaker contains an egg immersed in vinegar. Visitors observe carbon dioxide gas escaping from the shell as the calcium carbonate reacts with the vinegar.

Chemistry Is Colorful
Source Institutions
In "Chemistry is Colorful" learners explore different materials through paper chromatography.

Edible Glass
Source Institutions
In this activity, learners discover the principles of edible glass by making a supersaturated sugar solution.

It's a Gas!
Source Institutions
In this activity, learners explore two properties of gases: gases take up space and exert pressure. Learners assemble two flasks and a beaker, connecting them with stoppers and tubing.
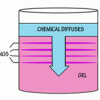
Liesegang Rings
Source Institutions
This display shows slow chemical reactions in colorful crystal formations known as Liesegang Rings. These reactions are similar to those forming the rings in agates.

Crocodiles
Source Institutions
Learners observe and compare the sizes of three toy “growing” crocodiles made from water-absorbent polymers. One is it its original state, dry, hard, and about 10cm long.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Crunch and Munch Lab
Source Institutions
In this activity, learners use three types of cheesy snacks--cheese balls, cheese puffs, and Cheetos--to learn about polymers.

Milk Makes Me Sick: Exploration of Lactose Intolerance
Source Institutions
Why does milk make some people sick? In this activity learners explore this question and explore the chemistry of milk, and our bodies!

What Does Spit Do?
Source Institutions
Some animals can swallow food whole, but humans have to chew. In this activity, learners will investigate what saliva does chemically to food before we even swallow.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

What's the Matter
Source Institutions
In this activity, learners identify different classes of matter based on physical properties.
