Search Results
Showing results 1 to 20 of 23

Matter on the Move
Source Institutions
Learners observe and conduct experiments demonstrating the different properties of hot and cold materials.

Invisible Ink Demonstration
Source Institutions
In this chemistry demonstration, learners will discover that phenolphthalein is a chemical that displays different colors depending on the acidity or basicity of the environment.

Why Doesn’t the Ocean Freeze?
Source Institutions
In this activity, learners explore how salt water freezes in comparison to fresh water.

Evaporation
Source Institutions
This three-part activity consists of an activity that groups of learners develop themselves, a given procedure, and an optional demonstration.

It's a Gas, Man
Source Institutions
In this activity, learners discover if carbon dioxide has an effect on temperature.

Amphibian Skin
Source Institutions
In this activity, learners explore the concept of permeability to better understand why amphibians are extremely sensitive to pollution.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Shrinkers
Source Institutions
In this hands-on activity, learners use heat to shrink samples of polystyrene plastic (#6 recycle code). Learners compare the size and shape of the plastic pieces before and after shrinking.

Nanoparticle Stained Glass
Source Institutions
In this activity/demo, learners are introduced to the connection between medieval stained glass artisans and nanotechnology.

Balloon in a Bottle
Source Institutions
In this physics activity (page 3 of the PDF), learners will see firsthand that air takes up space and has pressure by attempting to inflate a balloon inside of a bottle.

Rollin’ Rollin' Rollin'
Source Institutions
In this physics activity (page 12 of the PDF), learners explore potential and kinetic energy by rolling different sized marbles down an inclined plane.
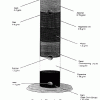
Daffy Density
Source Institutions
In this chemistry activity, learners explore density by using four solids and 6 liquids to create colorful, layered rows.

Canned Heat
Source Institutions
In this activity, learners explore how light and dark colored objects absorb the Sun's radiations at different rates.
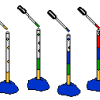
Liquid Layers
Source Institutions
Experiment with liquids of different densities and create liquid layers. For example, oil and water have different densities: oil floats on water because it is less dense than water.

Condensation
Source Institutions
In this activity, learners explore the process of condensation.
Let's Go Ice Fishing
Source Institutions
In this activity, learners are challenged to lift a floating ice cube out of a glass of water using just one end of a piece of string.

Melts in Your Bag, Not in Your Hand
Source Institutions
In this activity, learners use chocolate to explore how the Sun transfers heat to the Earth through radiation.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Powder Particulars
Source Institutions
In this introductory activity and demonstration, learners are introduced to the concept that different substances react chemically in characteristic ways.

Make a Salt Volcano (Lava Lite)
Source Institutions
This activity about density provides instructions for making a miniature "lava lite" with just salt, oil, water, and food coloring.
