Search Results
Showing results 21 to 40 of 90

My Angle on Cooling: Effects of Distance and Inclination
Source Institutions
In this activity, learners discover that one way to cool an object in the presence of a heat source is to increase the distance from it or change the angle at which it is faced.

Weather Stations: Temperature and Pressure
Source Institutions
In this activity, learners discover the relationship between temperature and pressure in the lower atmospheres of Jupiter and Earth.

Rubber Blubber Gloves
Source Institutions
In this experiment, learners work in pairs to create two gloves -- one that contains a layer of shortening (blubber) inside, and one that doesn't.

What Counts in Bounce
Source Institutions
In this activity learners compare the bounciness of warm and cold racquetballs to see if temperature makes a difference in how well they bounce.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.
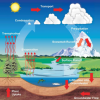
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.

Let's Dew It!
Source Institutions
From the Weather Watchers featured theme on the CYBERCHASE website. Learners will conduct experiments to discover how air temperature and humidity work together to make condensation, dew, and fog.

Make a Terrarium
Source Institutions
In this activity, learners make a miniature greenhouse or "terrarium" to explore the greenhouse effect.
Effects of Solar Radiation on Land and Sea
Source Institutions
In this activity, learners explore the different heating properties of soil and water.
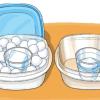
Save a Snowman
Source Institutions
In this activity, learners explore the concept of insulation by trying to keep Olaf the Snowman from melting. This activity encourages critical thinking and problem solving.

Try Your Hand at Nano
Source Institutions
This lesson focuses on two simple activities that younger learners can do to gain an appreciation of nanotechnology. First, learners measure their hands in nanometers.

What does Color have to do with Cooling?
Source Institutions
In this demonstration/experiment, learners discover that different colors and materials (metals, fabrics, paints) radiate different amounts of energy and therefore, cool at different rates.

Pie-Pan Convection
Source Institutions
It's difficult to see convection currents in any liquid that's undergoing a temperature change, but in this Exploratorium Science Snack, you can see the currents with the help of food coloring.
Cooling the Mummy's Tomb
Source Institutions
In this activity, learners conduct an experiment to help Pharaoh design a better insulated tomb.

Doghouse Design
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Doghouse Design) is a full inquiry investigation into absorption and reflection of radiant energy.

Avogadro's Bubbly Adventure
Source Institutions
In this activity on page 7 of the PDF, learners investigate the solubility of gas in water at different temperatures. This experiment will help learners determine if temperature affects solubility.

Conduction Countdown
Source Institutions
In this quick SciGirls activity (page 1 of the PDF under SciGirls Activity: Doghouse Design), learners will be introduced to the concept of thermal conductivity.

Temperature vs. Height: Soda Geyser Series #6
Source Institutions
In this activity, learners conduct a controlled experiment to examine how temperature will affect the height of a soda geyser.

Instant Ice Cream with a Dry Ice Bath
Source Institutions
In this chemistry meets cooking activity, learners make carbonated, vanilla ice cream using dry ice and denatured ethanol, which are both inexpensive and accessible.

Gas Model
Source Institutions
This highly visual model demonstrates the atomic theory of matter which states that a gas is made up of tiny particles of atoms that are in constant motion, smashing into each other.
