Search Results
Showing results 1 to 20 of 22

Latent Prints
Source Institutions
In this activity, learners examine fingerprints. Learners leave a hidden print on a surface and then make their own print detecting powder from graphite (found in pencils).

Mineral Scavenger Hunt
Source Institutions
In this activity, learners participate in a scavenger hunt, searching for and recognizing minerals and products that contain minerals.

Gas Production: Blow up a balloon!
Source Institutions
In this classic reaction, learners baking soda and vinegar in a soda bottle to produce carbon dioxide (CO2) gas. This gas inflates a balloon.

Kool Colors
Source Institutions
Learners investigate how temperature affects the rate of chemical reactions by observing how steel wool reacts with various types of Kool-Aid solutions at different temperatures.

Odors Aloft
Source Institutions
Learners smell balloons filled with different scents to guess what's inside. From this, they infer the presence and motion of scented molecules.

Density Rainbow
Source Institutions
In this activity, learners mix several sugar solutions to investigate the property of density. Each sugar solution has a different density and color of the rainbow.
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d
Penny for Your Thoughts
Source Institutions
In this activity, learners will explore how metals react with each other. They will see these metals change before their eyes as they coat a paperclip with the copper taken from a penny.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.
Plastic Milk: You can make plastic from milk
Source Institutions
In this activity (on page 2 of the PDF), learners make a plastic protein polymer from milk. Adding vinegar to milk causes the protein casein to solidify or curdle.
Cloudy Globs: Can You Make a White Gel From Two Clear Liquids?
Source Institutions
Using household materials, learners can make white gooey globs from clear solutions. Alum, dissolved in water, reacts with the hydroxide in ammonia to create aluminum hydroxide.

Cabbage Juice Indicator: Test the pH of household products
Source Institutions
Learners make their own acid-base indicator from red cabbage. They use this indicator to test substances around the house.
Choose Your Ooze
Source Institutions
During this activity, learners will make different versions of "ooze" using varied proportions of detergent and glue.

DNA Extraction
Source Institutions
Learners use a simple process to extract DNA from strawberries.

Egg Osmosis: A four day eggsperience!
Source Institutions
Eggs are placed in vinegar for one or two days to dissolve the shells. Then, learners place the eggs in water or corn syrup and observe them over a period of days.
Sticky Situation
Source Institutions
In this activity, learners separate the protein from milk and and use it to make their own glue.
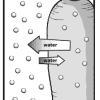
Bend a Carrot
Source Institutions
In this activity, learners investigate the process of osmosis by adding salt to a sealed bag of raw carrots and comparing it to a control.
