Search Results
Showing results 1 to 11 of 11

Matter on the Move
Source Institutions
Learners observe and conduct experiments demonstrating the different properties of hot and cold materials.

Watching Crystals Grow
Source Institutions
Learners will compare the growth rate and appearance of crystals forming on small rocks to those growing on miscellaneous objects. Learners will also investigate how temperature (warm vs.

Evaporation
Source Institutions
This three-part activity consists of an activity that groups of learners develop themselves, a given procedure, and an optional demonstration.
Cool Trees
Source Institutions
This warm weather activity introduces learners to the impact trees have on blocking the sun's heat and reducing temperature on the Earth's surface.

Insulation Station
Source Institutions
In this physical sciences activity, learners explore insulation. Leaners investigate how insulation can be used to slow down the conduction of heat from one side of a wall to the other.

Weather Stations: Temperature and Pressure
Source Institutions
In this activity, learners discover the relationship between temperature and pressure in the lower atmospheres of Jupiter and Earth.
Turning the Air Upside Down: Convection Current Model
Learners see convection currents in action in this highly visual demonstration. Sealed bags of colored hot or cold water are immersed in tanks of water.

Hot Stuff!
Source Institutions
In this activity, learners discover that sand is the major ingredient in glass.

Weather Stations: Winds
Source Institutions
In this activity, learners use a toaster to generate wind and compare the appliance's heat source to Jupiter's own hot interior. Learners discover that convection drives wind on Jupiter and on Earth.

Changing the Density of a Liquid: Heating and Cooling
Source Institutions
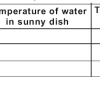
Learners investigate how the temperature of water affects its density.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.
