Search Results
Showing results 81 to 94 of 94

Dissolving Different Liquids in Water
Source Institutions
In this activity, learners add different liquids to water and apply their working definition of “dissolving” to their observations.

Avi's Sensational Salt Dough
Source Institutions
In this activity on page 5 of the PDF, learners mimic the process for making bricks. Learners shape and bake creations from a dough that is made from flour, salt, and water.

Cleaning Water with Dirt
Source Institutions
In this activity on page 7 of the PDF (Water in Our World), learners make their own water treatment systems for cleaning water.

Testing Vitamin C: Chemistry's Clear Solution
Source Institutions
In this activity on page 8 of the PDF, learners investigate vitamin C. Learners conduct a chemistry experiment to determine if Tang drink mix or orange juice contains more vitamin C.

Changing the Density of a Liquid: Heating and Cooling
Source Institutions
Learners investigate how the temperature of water affects its density.
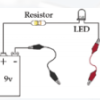
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.

Homework, Hogwarts Style
Source Institutions
In this activity on page 8 of the PDF (Behind the Scenes with Chemistry), learners make three of Harry Potter's essential school supplies: quills, ink, and color-changing paper.

Comparing the Amount of Acid in Different Solutions
Source Institutions
In this activity, learners use detergent solution to compare two solutions containing vinegar and cream of tartar.

Temperature Affects Dissolving
Source Institutions
Learners design their own experiment to compare how well cocoa mix dissolves in cold and hot water. They will see that cocoa mix dissolves much better in hot water. Adult supervision recommended.

Apples with Appeal
Source Institutions
In this activity, learners investigate why apples turn brown. Learners discover that lemon juice interferes with the reaction that causes the browning.

Changing the Density of an Object: Adding Material
Source Institutions
Learners see that a can of regular cola sinks while a can of diet cola floats. As a demonstration, bubble wrap is taped to the can of regular cola to make it float.

Photosynthesis and Transpiration
Source Institutions
In this activity on page 7 of the PDF (Plants—The Green Machines), learners examine the effects that light and air have on green plants.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.

Production of a Gas: Controlling a Chemical Reaction
Source Institutions
Learners mix vinegar and baking soda to produce a gas. With the addition of a bit of liquid soap, the gas becomes trapped in measurable bubbles.
