Search Results
Showing results 1 to 20 of 38

Density: Make a golf ball float!
Source Institutions
In this activity (on page 2 of the PDF), the learner places a golf ball between salt water and colored fresh water. The golf ball is not as dense as the saltwater.

Comparing the Density of an Object to the Density of Water
Source Institutions
Learners compare the weight of equal volumes of wax, water, and clay. Learners discover that since the wax weighs less than an equal volume of water, it is less dense than water and will float.

Defining Density
Source Institutions
In this introductory demonstration and activity, learners are introduced to the concept of density as they explore a rock and a wooden block in water.

Changing the Density of an Object: Changing Shape
Source Institutions
Learners will see that changing the shape of an object, like a clay ball, that is more dense than water, can affect whether the object will sink or float.

How Can Gravity Make Something Go Up?
Source Institutions
In this activity, learners use cheap, thin plastic garbage bags to quickly build a solar hot air balloon. In doing so, learners will explore why hot air rises.

Linear Functions: Mystery Liquids
Source Institutions
In this math lesson, learners analyze the density of liquids in order to explore linear functions.

Exploring How Liquids Behave
Source Institutions
Learners apply their knowledge from a previous study to identify different liquids--water, corn syrup, and vegetable oil.
Exploring Liquids
Source Institutions
Young learners investigate and observe the properties of three liquids -- water, vegetable oil, and corn syrup. They use their senses to collect data and ask and answer questions.

Comparing the Density of Different Liquids
Source Institutions
Learners carefully pour vegetable oil, water, and corn syrup in any order into a cup and discover that regardless of the order they are poured, the liquids arrange themselves in layers the same way.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.
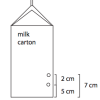
Pressing Pressure
Source Institutions
In this activity, learners compare water pressure at different depths. Learners discover that water pressure increases with depth.

The Amazing Water Trick
Source Institutions
Using two baby food jars, food coloring, and an index card, you'll 'marry' the jars to see how hot water and cold water mix.

Dancing Spaghetti
Source Institutions
In this chemistry activity, learners use spaghetti to explore density and chemical reactions.

Diving Submarine
Source Institutions
Learners use a commercially available toy to experiment with density. They fill a chamber in the toy submarine with baking powder and release it into a tank of water.

Test Density with a Supersaturated Solution
Source Institutions
Learners create three solutions with different levels of salinity. They compare the density of these solutions by coloring them and layering them in a clear plastic cup and in a soda bottle.
Milli's Super Sorting Challenge
Source Institutions
In this activity, learners separate materials based on their special properties to mimic the way recyclables are sorted at recycling centers.

Balloon Flinker
Source Institutions
In this activity, learners make a helium balloon "flink"--neither float away nor sink to the ground. Use this activity to introduce physics concepts related to gravity, density, and weight.

Color Splash
Source Institutions
In this activity, learners mix water, cooking oil, and liquid food coloring to create beautiful colored designs in a cup. Use this activity to explore liquid density and solubility.

Energetic Water
Source Institutions
In this activity, learners explore how hot and cold water move. Learners observe that temperature and density affect how liquids rise and fall.
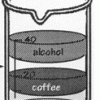
Layers of Liquids
Source Institutions
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.
