Search Results
Showing results 1 to 16 of 16
Charge Challenge
Source Institutions
In this activity, learners explore how objects can have positive, negative, or neutral charges, which attract, repel and move between objects.

Polyatomic Ion Bingo
Source Institutions
This activity was designed for blind learners, but all types of learners can play this game to learn about the major polyatomic ions (an ion that consists of two different elements).

Indicating Electrolysis
Source Institutions
In this activity, learners build a simple electrolysis device. Then learners use an indicating solution to visualize hydrogen and oxygen molecules in water.
Ionic Bonding Puzzle Lab
Source Institutions
In this activity, learners create models of ionic compounds and observe the chemical formula of binary molecules they have created.

Electroplating
Source Institutions
In this activity, learners electrically plate zinc onto brass objects.

Can You Copperplate?
Source Institutions
In this activity, learners explore chemical engineering and how the processes of chemical plating and electroplating have impacted many industries.

Squidgy Slime
Source Institutions
In this chemistry activity, learners transform two ingredients (4% polyvinyl alcohol solution and 4% borax solution) into gooey slime.

Having a Gas with Water
Source Institutions
In this activity, learners construct a simple electrolysis device. With this device, learners can decompose water into its elemental components: hydrogen and oxygen gas.

Defining Dissolving
Source Institutions
In this introductory activity, learners discover that sugar and food coloring dissolve in water but neither dissolves in oil.

Solubility Test
Source Institutions
In this activity, learners apply a dissolving test to known crystals to identify the unknown. Since the unknown is chemically the same as one of the known crystals, it should dissolve similarly.

Dusted!
Source Institutions
Learners press their fingertip onto a clean Plexiglas sheet. The fingerprints are then revealed as learners dust over the print with fingerprint powder.
Salts & Solubility
Source Institutions
In this online interactive simulation, learners will add different salts to water and then watch the salts dissolve and achieve a dynamic equilibrium with solid precipitate.

Conductivity Meter
Source Institutions
In this activity, learners build a simple qualitative conductivity tester with a battery, bulb and foil.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.

Coral, Carbon Dioxide and Calcification
Source Institutions
In this group activity, learners act out key stages of the "ocean carbon cycle" (also known as the "carbonate buffer system") through motions, rearranging blocks and team tasks.
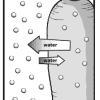
Bend a Carrot
Source Institutions
In this activity, learners investigate the process of osmosis by adding salt to a sealed bag of raw carrots and comparing it to a control.
