Search Results
Showing results 1 to 20 of 32

A Little Drop of Water: Cohesion
Source Institutions
Learners explore water's property of cohesion through two investigations.

Collect Oxygen Over Water
Source Institutions
In this activity, learners use a pneumatic trough (see related activity) to generate and collect pure oxygen.

A Recipe for Air
Learners use M&Ms® (or any other multi-color, equally-sized small candy or pieces) to create a pie graph that expresses the composition of air.

Vital Signs
Source Institutions
This activity (on pages 16-23) lets learners measure each other's vital signs—the signs that help doctors understand what's going on in a patient body.
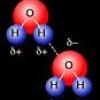
Cohesion Coin
Source Institutions
In this activity about the property of water (page 6 of the PDF), learners use a coin to demonstrate cohesion between water molecules, exploring the molecular forces that allow water molecules to "

Fast Rusting
Source Institutions
In this activity, learners conduct an experiment to find out if steel wool will weigh more or less when it is burned. Learners will explore the effects of oxidation and rusting on the steel wool.
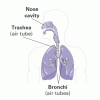
Heart and Lungs
Source Institutions
In this environmental health activity, learners investigate their breathing and pulse rates, and learn how these measurements are affected by physical activity.

What's In Your Breath?
Source Institutions
In this activity, learners test to see if carbon dioxide is present in the air we breathe in and out by using a detector made from red cabbage.
Carbon Dioxide Removal
Source Institutions
In this experiment using sprigs of Elodea, learners will observe a natural process that removes carbon dioxide (CO2) from Earth's atmosphere.

Indicating Electrolysis
Source Institutions
In this activity, learners build a simple electrolysis device. Then learners use an indicating solution to visualize hydrogen and oxygen molecules in water.

What's in the Water?: Biotic and Abiotic Elements in Aquatic Ecosystems
Source Institutions
In this investigation learners explore the differences between, and interdependence of, living and nonliving elements in a water ecosystem.

What Color is Your Air Today?
Learners develop awareness and understanding of the daily air quality using the Air Quality Index (AQI) listed in the newspaper or online.

Respiration Relay
Source Institutions
In this physical activity, groups of learners act as blood cells traveling through the circulatory system.

Earth Atmosphere Composition
Source Institutions
In this activity, learners use rice grains to model the composition of the atmosphere of the Earth today and in 1880. Learners assemble the model while measuring percentages.

Lifting Lemon
Source Institutions
In this physics demonstration, learners will be surprised when a lemon slice appears to magically levitate within a pint glass.
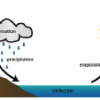
The Water Cycle
Source Institutions
Did you know that the water we use today is the same water found on Earth millions of years ago? The Earth constantly uses and recycles water in a process called the water cycle.

Indicating Electrolysis
Source Institutions
Electrolysis is the breakdown of water into hydrogen and oxygen. This Exploratorium activity allows learners to visualize the process with an acid-based indicator.

Rocket Science
Source Institutions
Learners create a small explosion by collecting hydrogen and oxygen gas together and squeezing them into a flame.

Breaking Up with Combustion
Source Institutions
This activity teaches combustion as the interaction of a fuel source and oxygen.

Battling for Oxygen
Working in groups, learners model the continuous destruction and creation of ozone (O3) molecules, which occur in the ozone layer.
