Search Results
Showing results 1 to 17 of 17
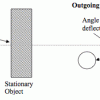
Where Will It Go?
Source Institutions
In this activity, learners predict where a ball will go after it bounces off another object. Learners discover that the motion of objects is predictable based on laws of motion.
Charge Challenge
Source Institutions
In this activity, learners explore how objects can have positive, negative, or neutral charges, which attract, repel and move between objects.

Magnet Mania
Source Institutions
In this activity, learners explore the relationship between electric charges and magnetic fields.

What Shape Is It?
Source Institutions
In this activity, learners determine the shape of an unseen object by bouncing a ball off the object.

Forgotten Genius
Source Institutions
This series of chemistry stations is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Each of the six stations features either a chemical or p

Coral, Carbon Dioxide and Calcification
Source Institutions
In this group activity, learners act out key stages of the "ocean carbon cycle" (also known as the "carbonate buffer system") through motions, rearranging blocks and team tasks.

Antigen-Antibody Testing: A Visual Simulation or Virtual Reality
Source Institutions
In this biology activity, learners use plastic pipettes to cut wells into the solid gel layer of agar in petri dishes and place solutions of simulated antigen and antibody preparations into the wells.
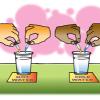
Molecules in Motion
Source Institutions
In this activity, learners add food coloring to hot and cold water to see whether heating or cooling affects the speed of water molecules.

Spectroscope
Source Institutions
In this activity (posted on March 12, 2011), learners follow the steps to construct a spectroscope, a tool used to analyze light and color.

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.

The Carbon Cycle and its Role in Climate Change: Activity 1
Source Institutions
In this activity (on page 1), learners role play as atoms to explore how atoms can be rearranged to make different materials.

Indicating Electrolysis
Source Institutions
In this activity, learners build a simple electrolysis device. Then learners use an indicating solution to visualize hydrogen and oxygen molecules in water.

Liquid Crystal Thermometers
Source Institutions
In this activity, learners explore liquid crystal thermometers to observe how heat flows by conduction, convection, radiation, and evaporation.
Rutherford's Enlarged: A Content-Embedded Activity to Teach about Nature of Science
Source Institutions
This paper describes a working-model demonstration of Ernest Rutherford's 1911 experiment about the nature of atoms.

How Small Can You Cut?
Source Institutions
In this lesson, learners cut paper into very small pieces to explore the small size of quarks, the smallest thing we know of on Earth.
Invisible Investigations
Source Institutions
Using indirect observational methods, learners distinguish between charged and uncharged objects.

The Ups and Downs of Thermometers
Source Institutions
In this activity, learners examine the parts of a thermometer. After placing a thermometer in hot and cold water, learners look at molecular model animations of the liquid in a thermometer.
