Search Results
Showing results 1 to 19 of 19

Metal Reactions
Source Institutions
This is written as a static display, but can easily be adapted to a hands-on experiment for learners to conduct.

Wheat Evolution: Sedimentation Testing
Source Institutions
In this activity (Page 30 of PDF), learners investigate the evolution of wheat by conducting sedimentation tests on different flours.

Invisible Ink Demonstration
Source Institutions
In this chemistry demonstration, learners will discover that phenolphthalein is a chemical that displays different colors depending on the acidity or basicity of the environment.

Fireworks!
Source Institutions
In this chemistry lab activity, learners model the colors of fireworks by burning metallic solutions in a flame and observing the different colors produced.

Phase Changes
Source Institutions
Learners observe a sealed test tube containing a small amount of solid stearic acid.

Corals and Chemistry
Source Institutions
In this activity, learners investigate how increased carbon dioxide (CO2) emissions from the burning of fossil fuels is changing the acidity (pH) of the ocean and affecting coral reefs and other marin

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.
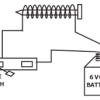
How Do We Convert Electrical Energy into Mechanical Energy?
Source Institutions
In this activity, learners make an electromagnet motor to demonstrate the most basic method of changing electrical energy into mechanical energy.

Sink It
Source Institutions
Learners classify a variety of objects by their characteristics. They then design an experiment to determine which objects float or sink in water and add this characteristic to their classification.
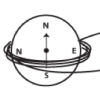
How Do We Convert Mechanical Energy into Electrical Energy?
Source Institutions
In this activity, learners use a compass, powerful magnet, and copper magnet wire to build a special generator known as a dynamo.

What is a “Convection Cell”?
Source Institutions
In this demonstration, learners can observe a number of small convection cells generated from a mixture of aluminum powder and silicon oil on a hot plate.
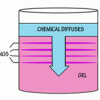
Liesegang Rings
Source Institutions
This display shows slow chemical reactions in colorful crystal formations known as Liesegang Rings. These reactions are similar to those forming the rings in agates.

Bend That Bar
Learners play the role of materials engineers as they test the flexibility of different materials.

Food for the Brain
Source Institutions
In this activity, learners dissect a piece of pizza to learn about nutrients important for health.

Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.
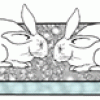
Acid Rain Eats Stone!
Source Institutions
This display shows the dangers of acid rain on buildings and other structures as two concrete bunny rabbits are disintegrated by sulfuric acid. Learners scrape chalk onto the concrete bunnies.

Global Climate Change and Sea Level Rise
Source Institutions
In this activity, learners practice the steps involved in a scientific investigation while learning why ice formations on land (not those on water) will cause a rise in sea level upon melting.

