Search Results
Showing results 1 to 20 of 32

Soap: Sometimes oil and water do mix!
Source Institutions
In this activity (on page 2 of PDF), learners mix oil and water. Then, they add soap and observe what changes! The activity demonstrates how oil and water don't mix, except when soap is added.

Density: Make a golf ball float!
Source Institutions
In this activity (on page 2 of the PDF), the learner places a golf ball between salt water and colored fresh water. The golf ball is not as dense as the saltwater.

Odors Aloft
Source Institutions
Learners smell balloons filled with different scents to guess what's inside. From this, they infer the presence and motion of scented molecules.

Lava Lamps
Source Institutions
Learners observe working lava lamps to understand how they work (included in PDF link).

The Proof is in the Powder
Source Institutions
In this activity, learners will design a way to identify a powder found at a crime scene by comparing it with known powders, with the goal of solving a crime.

DNA Extraction
Source Institutions
Learners use a simple process to extract DNA from strawberries.
Bend a Carrot
Source Institutions
In this activity, learners investigate the process of osmosis by adding salt to a sealed bag of raw carrots and comparing it to a control.

In the Toilet
Source Institutions
This activity explores the basic workings of a siphon, which is the core technology that makes toilets work.

Phase Changes
Source Institutions
Learners observe a sealed test tube containing a small amount of solid stearic acid.
Ice Cube Painting
Source Institutions
In this activity, learners "draw" with frozen tempera paint. The ice cubes are prepared the day before by placing watered down tempera paint and popsicle sticks in ice cube trays.
Salting Out
Source Institutions
In this activity, learners create a mixture of water, alcohol and permanent marker ink, and then add salt to form a colored alcohol layer on top of a colorless water layer.

Pot-in-Pot Refrigeration
Source Institutions
In this activity (on page 2 of PDF), learners create a low-tech refrigerator that requires no electricity to keep food from spoiling.

To Dye For
Source Institutions
Learners add two dyes to mineral oil and water, and then compare their miscibility (how well they mix) in each.

Cool It!
Source Institutions
Learners make a refrigerator that works without electricity. The pot-in-pot refrigerator works by evaporation: a layer of sand is placed between two terra cotta pots and thoroughly soaked with water.

Miscibility
Source Institutions
Learners observe a bottle containing water and oil. They are invited to pick up the bottle and mix the contents together.

Currently Working
Source Institutions
Learners test solutions of water, sugar, salt, and hydrochloric acid for electrical conductivity. They immerse leads from a lighting device (a battery pack connected to an LED) into each solution.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.
Cloudy Globs: Can You Make a White Gel From Two Clear Liquids?
Source Institutions
Using household materials, learners can make white gooey globs from clear solutions. Alum, dissolved in water, reacts with the hydroxide in ammonia to create aluminum hydroxide.
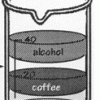
Layers of Liquids
Source Institutions
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.
