Search Results
Showing results 1 to 20 of 45

Rusty Penny
Source Institutions
In this easy chemistry activity, learners submerge pennies in different liquids (water, lemon juice, vinegar, liquid hand soap, salt water, and baking soda mixed with water) to observe which best clea
Mercury in the Environment
Source Institutions
In this environmental science lesson, learners will examine the dangers of mercury and how humans contribute to growing mercury emissions on Earth.

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.
Investigating Density Currents
Source Institutions
In this lab activity, learners explore how to initiate a density current. Learners measure six flasks with different concentrations of salt and water (colored blue).

Watching Crystals Grow
Source Institutions
Learners will compare the growth rate and appearance of crystals forming on small rocks to those growing on miscellaneous objects. Learners will also investigate how temperature (warm vs.
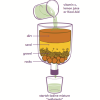
Water Clean-up
Source Institutions
This is an activity (located on page 3 of the PDF under Water Clean-up Activity) about the use of reduction agents to decontaminate ground water.

What's Hiding in the Air?: Acid Rain Activity
As a model of acid rain, learners water plants with three different solutions: water only, vinegar only, vinegar-water mixture.

Radioactive Decay of Candium
Source Institutions
In this simulation, learners use M&M™ candy to explore radioactive isotope decay.

Composting
Source Institutions
In this environmental science activity, learners research what is essential for plant life and the necessary components of soil to support plants.

How Big is Small
Source Institutions
In this classic hands-on activity, learners estimate the length of a molecule by floating a fatty acid (oleic acid) on water.

Does Sunscreen Protect My DNA?
Source Institutions
In this laboratory experiment, learners explore how effectively different sunscreens protect yeast cells from damage caused by ultraviolet (UV) radiation.
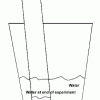
Percentage of Oxygen in the Air
Source Institutions
In this activity, learners calculate the percentage of oxygen in the atmosphere by using steel wool's ability to rust.
What's So Special about Water: Absorption
Source Institutions
In this activity about water's cohesive and adhesive properties and why water molecules are attracted to each other, learners test if objects repel or absorb water.

Crystal Gardens
Source Institutions
In this activity, which requires adult supervision, learners get to explore the awesome power of chemistry.

Gravestone Weathering
Source Institutions
In this activity (located on pages 9-14 of PDF), learners visit a cemetery to examine the distinguishing characteristics of rock weathering.

Aerogel
Source Institutions
This activity/demo introduces learners to aerogel, a glass nanofoam. Learners discover how aerogel is made and how well it insulates as well as learn about aerogel's other unique properties.

Avogadro's Bubbly Adventure
Source Institutions
In this activity on page 7 of the PDF, learners investigate the solubility of gas in water at different temperatures. This experiment will help learners determine if temperature affects solubility.

Cook Up a Comet
Source Institutions
In this activity (on page 5 of PDF), learners use dry ice and household materials to make scientifically accurate models of comets.

The Carbon Cycle and its Role in Climate Change: Activity 1
Source Institutions
In this activity (on page 1), learners role play as atoms to explore how atoms can be rearranged to make different materials.

Candy Chemosynthesis
Source Institutions
In this activity, groups of learners work together to create edible models of chemicals involved in autotrophic nutrition.
