Search Results
Showing results 1 to 20 of 30

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.

Fruity Electricity
Source Institutions
In this activity, Frankenstein's lab is running out of electricity! Learners use fruit to help Igor find a temporary source of energy to turn on a light.

Build a Battery
Source Institutions
Learners make a simple battery out of "sandwiches" of aluminum foil, pennies, and a salt water-soaked paper towel.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.

Forms of Carbon
Source Institutions
In this activity, educators can demonstrate how the nanoscale arrangement of atoms dramatically impacts a material’s macroscale behavior.

Biobarcodes: Antibodies and Nanosensors
Source Institutions
In this activity/demo, learners investigate biobarcodes, a nanomedical technology that allows for massively parallel testing that can assist with disease diagnosis.
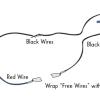
Neural Network Signals
Source Institutions
In this activity, learners create an electrical circuit and investigate how some dissolved substances conduct electricity.

Make Your Own Batteries!
Source Institutions
This activity (on page 3 of the PDF under GPS: Body Electricity Activity) is a full inquiry investigation into conductivity.

Harvesting Chemicals from a Battery
Source Institutions
In this activity, learners take apart a used zinc-carbon dry cell battery.

Shake and Make: Charge Recognition
Source Institutions
In this activity (page 10), learners explore how molecules self-assemble according to forces of attraction and repulsion.
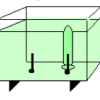
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.
Milli's Super Sorting Challenge
Source Institutions
In this activity, learners separate materials based on their special properties to mimic the way recyclables are sorted at recycling centers.

Atoms and Matter (K-2)
Source Institutions
In this activity, learners explore atoms as the smallest building blocks of matter. With adult help, learners start by dividing play dough in half, over and over again.

Can You Copperplate?
Source Institutions
In this activity, learners explore chemical engineering and how the processes of chemical plating and electroplating have impacted many industries.

Investigating and Using Biomass Gases
Source Institutions
In this activity, learners will be introduced to biomass gasification and will generate their own biomass gases.

Indicating Electrolysis
Source Institutions
Electrolysis is the breakdown of water into hydrogen and oxygen. This Exploratorium activity allows learners to visualize the process with an acid-based indicator.
Properties of Metals
Source Institutions
In this activity, learners explore the properties of metals at four stations. The stations include A) Magnetism and Breakfast Cereal; B) Conductivity of Metals; C) Alloys; and D) Metal Plating.

Home Molecular Genetics
Source Institutions
In this activity, learners extract DNA from their own cheek cells, then create a rudimentary DNA profile similar to those seen on crime scene dramas.

Exploring the Nanoworld with LEGO Bricks: Structure-Property Relationships at the Nanoscale
Source Institutions
In this activity (pages 32-41), learners learn how the atomic and molecular arrangement of matter are related to physical properties.

Density Intensity
Source Institutions
In this activity on page 12 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners examine how recyclable materials are separated by various properties at recycling centers.
