Search Results
Showing results 21 to 40 of 45

What is in the Water?
Source Institutions
In this activity, learners use open inquiry to learn about the process of science as well as gain experience regarding the Law of Conservation of Mass, dissolution, and density.

Layered Liquids: Chemistry You Can Drink
Source Institutions
In this chemistry activity (on page 2 of the PDF), learners make a layered drink with liquids of different densities.

Fly a Hot-Air Balloon
Source Institutions
Learners assemble a hot-air balloon from tissue paper. The heated air (from a heat gun) inside the balloon is less dense than the surrounding air and causes the balloon to float.
Daffy Density
Source Institutions
In this chemistry activity, learners explore density by using four solids and 6 liquids to create colorful, layered rows.
Floating Golf Ball
Source Institutions
Visitors observe a graduated cylinder with a golf ball floating about halfway in liquid. The bottom half of the cylinder contains a concentrated solution of salt.

Submarine: Lift Bag Lander
Source Institutions
In this activity (on page 4), learners create a submarine using a plastic sandwich bag. This is a fun way to learn about buoyancy and how captured gas can cause objects to float.
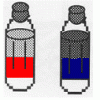
Oil and Soap
Source Institutions
Learners investigate the properties of the liquids in two bottles. One contains layers of oil and water, and one contains oil, water, and soap.
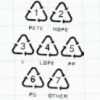
Sink or Swim?
Source Institutions
In this activity, learners identify different plastics in a mystery bag. Learners discover that plastics are classified #1 through #7.

Conversation Piece
Source Institutions
Focus sound through a balloon! In this Exploratorium activity, you'll use dry ice to create a balloon that's a sound lens.

Using Color to See How Liquids Combine
Source Institutions
Learners add different liquids (water, salt water, alcohol, and detergent solution) to water and observe the different ways the different liquids combine with water.

Salt Crystal Garden
Source Institutions
In this activity, learners will explore saturated solutions and discover how crystals form.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.

Oil Spill Cleanup
This hands-on experiment will provide learners with an understanding of the issues that surround environmental cleanup.

Solar Convection
Source Institutions
In this activity, learners add food coloring to hot and cold water in order to see how fluids at different temperatures move around in convection currents.

Density Rainbow
Source Institutions
In this activity, learners mix several sugar solutions to investigate the property of density. Each sugar solution has a different density and color of the rainbow.

Density Rainbows
Source Institutions
In this activity, learners explore the concept of density by pouring 5 different liquids into a jar. Food coloring is added if needed to give each liquid a distinct color.

Bubble Suspension
Source Institutions
In this activity, learners observe as soap bubbles float on a cushion of carbon dioxide gas. Learners blow bubbles into an aquarium filled with a slab of dry ice.

Challenge: Microgravity
Source Institutions
In this activity about the circulatory system and space travel (on page 38 of the PDF), learners use water balloons to simulate the effects of gravity and microgravity on fluid distribution in the bod

Kimchee Fermentation Chamber
Source Institutions
Learners make kimchee or sauerkraut, which is really just fermented cabbage, in a 2-liter plastic bottle.
