Search Results
Showing results 41 to 60 of 92

Temperature vs. Height: Soda Geyser Series #6
Source Institutions
In this activity, learners conduct a controlled experiment to examine how temperature will affect the height of a soda geyser.

Instant Ice Cream with a Dry Ice Bath
Source Institutions
In this chemistry meets cooking activity, learners make carbonated, vanilla ice cream using dry ice and denatured ethanol, which are both inexpensive and accessible.

Gas Model
Source Institutions
This highly visual model demonstrates the atomic theory of matter which states that a gas is made up of tiny particles of atoms that are in constant motion, smashing into each other.

Sinking Water
Source Institutions
In this experiment, learners float colored ice cubes in hot and cold water.
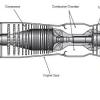
Jet Propulsion
Source Institutions
In this two-part activity, learners work in pairs to examine the four basic stages of a turbine engine.

Differing Densities: Fresh and Salt Water
Source Institutions
In this activity, learners visualize the differences in water density and relate this to the potential consequences of increased glacial melting.

Temperature Tactics
Source Institutions
In this activity, learners explore the devices used over time to measure changes in temperature.

Feel the Heat
Source Institutions
In this design challenge activity, learners design and build a solar hot water heater. Their goal is to create a heater that yields the highest temperature change.

Chocolate (Sea Floor) Lava
Source Institutions
In this edible experiment, learners pour "Magic Shell" chocolate into a glass of cold water. They'll observe as pillow shaped structures form, which resemble lavas on the sea floor.

How does the Atmosphere keep the Earth Warmer?
Source Institutions
In this activity, learners simulate the energy transfer between the earth and space by using the light from a desk lamp desk lamp with an incandescent bulb and a stack of glass plates.
Physical Change
Source Institutions
In this activity, learners use heat to separate zinc and copper in a penny. This experiment demonstrates physical properties and how physical change (phase change) can be used to separate matter.
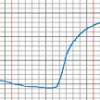
The Daily Ups and Downs
Source Institutions
In this activity, learners graph 48 hourly air temperatures from a local weather observation site and observe the diurnal temperature variations.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Meltdown
Source Institutions
In this activity, learners heat ice and water of the same temperature to get a hands-on look at phase changes. This is an easy and inexpensive way to introduce states of matter and thermodynamics.

House Warming
Source Institutions
In this physical sciences activity, learners explore how passive solar design increases energy efficiency.
What's the Angle?
Source Institutions
This activity helps learners understand how the angle of the Sun affects temperatures around the globe.

That Sinking Feeling
Source Institutions
In this quick activity, learners observe how salinity and temperature affect the density of water, to better understand the Great Ocean Conveyor.

Cold Metal
Source Institutions
In this activity, learners discover that our hands are not reliable thermometers.

Burn a Peanut
Source Institutions
In this activity, learners burn a peanut, which produces a flame that can be used to boil away water and count the calories contained in the peanut.

Give and Take
Source Institutions
In this activity, learners explore liquid crystals, light and temperature. Using a postcard made of temperature-sensitive liquid crystal material, learners monitor temperature changes.
