Search Results
Showing results 41 to 60 of 115

Chemistry Is Colorful
Source Institutions
In "Chemistry is Colorful" learners explore different materials through paper chromatography.

Acid Rain
Source Institutions
In this chemistry demonstration, acid rain is simulated in a petri dish.

Edible Glass
Source Institutions
In this activity, learners discover the principles of edible glass by making a supersaturated sugar solution.

Nanoparticle Stained Glass
Source Institutions
In this activity/demo, learners are introduced to the connection between medieval stained glass artisans and nanotechnology.

Seas in Motion
Source Institutions
In this outdoor, beach activity, learners use tennis balls, water balloons and other simple devices to investigate the movement of waves and currents off a sandy beach.
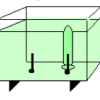
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.

Potato Power
Source Institutions
Learners combine hydrogen peroxide with three different forms of potato: raw chunks, ground chunks, and boiled chunks.

Currently Working
Source Institutions
Learners test solutions of water, sugar, salt, and hydrochloric acid for electrical conductivity. They immerse leads from a lighting device (a battery pack connected to an LED) into each solution.

Rock Bottoms
Source Institutions
Learners add acid rain (nitric acid) to two cups that represent lakes. One cup contains limestone gravel and the other contains granite gravel.
Magnetism
Source Institutions
In this activity, learners explore the principles of magnetism by observing how various materials interact with magnets and superconductors.
Foam Peanuts
Source Institutions
Learners compare the properties and solubilities of Styrofoam (TM), ecofoam packing peanuts, and popcorn. First, the solubility of each substance is tested in water.

It's a Gas!
Source Institutions
In this activity, learners explore two properties of gases: gases take up space and exert pressure. Learners assemble two flasks and a beaker, connecting them with stoppers and tubing.

Nature of Dye
Source Institutions
"Nature of Dye" allows participants to create their own dyes and art while exploring how chemicals interact and how these interactions can have real-world applications.

Food Forensics: A Case of Mistaken Identity
Source Institutions
This lesson is designed to serve as an introduction to the immune system. It can stand alone or it can lead into further studies of the immune system.

Making Metals Strong
Source Institutions
In this activity, learners demonstrate the effects of cold-working (strain-hardening) and annealing. Learners discover that these processes change the load that a wire can support.

Float Your Boat
Source Institutions
In this physics activity, learners will explore buoyancy.

Think Fast!: Just How Quick Are You?
Source Institutions
This is an activity about reaction times. Just how quickly must an NHL goalie respond to save a shot, and how does your reaction time compare?

Burning Issues
Source Institutions
Learners use a candle to investigate the products of combustion. When a glass rod is held over a lit candle, the candle flame deposits carbon on the rod.

Balloon in a Flask
Source Institutions
Learners observe a flask with a balloon attached over the mouth and inverted inside the flask.
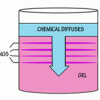
Liesegang Rings
Source Institutions
This display shows slow chemical reactions in colorful crystal formations known as Liesegang Rings. These reactions are similar to those forming the rings in agates.
