Search Results
Showing results 1 to 17 of 17

Exploring Materials: Liquid Crystals
Source Institutions
In this activity, learners discover that the way a material behaves on the macroscale is affected by its structure on the nanoscale.

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.

Rubber Band Thermodynamics
Source Institutions
In this demonstration, learners explore the thermal properties of rubber. Learners investigate whether a rubber band contracts or expands when heated.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Lava Lamps
Source Institutions
Learners observe working lava lamps to understand how they work (included in PDF link).

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).

Aerogel
Source Institutions
This activity/demo introduces learners to aerogel, a glass nanofoam. Learners discover how aerogel is made and how well it insulates as well as learn about aerogel's other unique properties.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Plastics the Second Time Around
Source Institutions
In this activity, learners test and compare the physical properties of thermoplastic polymers. Learners compare different plastics based on their color, degree of transparency, texture, and density.

Nuclear Fusion
Source Institutions
This simple and engaging astronomy activity explains nuclear fusion and how radiation is generated by stars, using marshmallows as a model.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Lifting Lemon
Source Institutions
In this physics demonstration, learners will be surprised when a lemon slice appears to magically levitate within a pint glass.

Magical Match
Source Institutions
In this demonstration, learners will be "wowed" as three matches burn to form a triangular pyramid shape and "magically" rise off the table.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
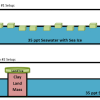
Causes and Effects of Melting Ice
Source Institutions
In this activity, learners explore the concept of density-driven currents (thermohaline circulation) and how these currents are affected by climate change.

That's the Way the Ball Bounces: Level 3
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.
