Search Results
Showing results 1 to 18 of 18
Monster Mallows
Source Institutions
In this activity, learners explore how ordinary marshmallows expand when heated in a microwave.

Underwater Fireworks
Source Institutions
In this activity, learners investigate diffusion by creating underwater "fireworks" using food coloring, oil and water.
Heat Speeds Up Reactions
Source Institutions
In this activity, learners investigate the effect of heat on a reaction.

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.

Shrinky Dinks
Source Institutions
Heat makes some materials expand, and it makes others shrink.

Changing Colors
Source Institutions
Learners experiment with a commercially available liquid-crystal coaster. They warm the material with their hands for varying lengths of time and observe the changing colors that result.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

The Amazing Water Trick
Source Institutions
Using two baby food jars, food coloring, and an index card, you'll 'marry' the jars to see how hot water and cold water mix.

Full of Hot Air: Hot Air Balloon Building
Source Institutions
In this activity, learners create a model of a hot air balloon using tissue paper and a hairdryer. Educators can use this activity to introduce learners to density and its role in why things float.

Mixing and Unmixing in the Kitchen
Source Institutions
In this chemistry investigation, learners combine common cooking substances (flour, baking powder, sugar, salt, pepper, oil, water, food coloring) to explore mixtures.

Pepper Scatter
Source Institutions
In this quick activity, learners break the tension that happens when water develops a "skin." Learners use water, pepper and some soap to discover the wonders of surface tension—the force that attract

Balloon in a Bottle
Source Institutions
In this physics activity (page 3 of the PDF), learners will see firsthand that air takes up space and has pressure by attempting to inflate a balloon inside of a bottle.

Film Canister Rocket
Source Institutions
In this activity, learners construct and launch rockets using simple materials and their understanding of chemical reactions.
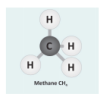
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Stained Glass Glue
Source Institutions
In this activity on page 6 of the PDF, learners use glue instead of glass to create artwork that can be hung in a window.

Mystery Shapes
Source Institutions
In this activity, learners describe an object they can’t see. After someone picks outs a few mystery objects and places them in a pillowcase, learners will investigate using only their hands.

Exploring Forces: Gravity
Source Institutions
In this nanoscience activity, learners discover that it's easy to pour water out of a regular-sized cup, but not out of a miniature cup.
