Search Results
Showing results 1 to 20 of 81

Drop Shape
Source Institutions
In this activity, learners get a closer look at the shape of a drop of water and a drop of oil. Learners first drip water onto wax paper and examine the shape of separate drops from a side view.

Look-alike Liquids
Source Institutions
Learners add drops of four liquids (water, alcohol, salt water, and detergent solution) to different surfaces and observe the liquids' behavior.

How Much Water is in that Cloud?
Source Institutions
In this activity, learners working in pairs saturate a cotton ball using water drops from an eyedropper to demonstrate the high water capacity of clouds.

Colors Collide or Combine
Source Institutions
Learners place multiple M&M's in a plate of water to watch what happens as the candies dissolve.

Which Powder is It?
Source Institutions
In this chemistry challenge, learners identify an unknown white powder by comparing it with common household powders.

Plant Power
Source Institutions
In this chemistry challenge, learners identify which plants have the enzyme "catalase" that breaks hydrogen peroxide into water and oxygen.

Exploring How Liquids Behave
Source Institutions
Learners apply their knowledge from a previous study to identify different liquids--water, corn syrup, and vegetable oil.

Hot and Cold
Source Institutions
In this chemistry challenge, learners discover that many chemical reactions involve heat loss or gain.

The Liquid Rainbow
Source Institutions
Learners are challenged to discover the relative densities of colored liquids to create a rainbow pattern in a test tube.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.
Exploring Liquids
Source Institutions
Young learners investigate and observe the properties of three liquids -- water, vegetable oil, and corn syrup. They use their senses to collect data and ask and answer questions.

Polymers are Chains (K-2)
Source Institutions
In this activity, learners make a paper model of a polymer, then make Silly Putty, an actual polymer.
Mix and Match
Source Institutions
In this optics activity, learners explore color by examining color dots through colored water and the light of a flashlight.
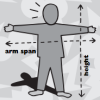
Are you a Square or a Rectangle?
Source Institutions
In this activity, learners investigate whether more people are squares or rectangles. People with similarly sized heights and arm spans are classified as squares.
Water Filter
Source Institutions
In this engineering activity, challenge learners to invent a water filter that cleans dirty water.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.

Mysterious M&M's
Source Institutions
Learners place an M&M candy in water and observe what happens. The sugar-and-color coating dissolves and spreads out in a circular pattern around the M&M.

Color Changes with Acids and Bases
Source Institutions
Learners mix a variety of substances with red cabbage juice. The juice changes color to indicate whether each substance is an acid or a base.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.
