Search Results
Showing results 1 to 12 of 12

Say Cheese!
Source Institutions
Create a chemical reaction that makes cheese! This hands-on activity demonstrates that molecules and atoms are tiny particles that make up everything around us.

Color Changes with Acids and Bases
Source Institutions
Learners mix a variety of substances with red cabbage juice. The juice changes color to indicate whether each substance is an acid or a base.

Neutralizing Acids and Bases
Source Institutions
Learners use their knowledge of color changes with red cabbage indicator to neutralize an acidic solution with a base and then neutralize a basic solution with an acid.

Acids & Bases
Source Institutions
In this activity, learners test the pH of safe liquids available at home by creating a pH indicator from mashed blueberries.

Hot Stuff!: Creating and Testing for Carbon Dioxide
In this demonstration, learners observe vinegar and baking soda reacting to form carbon dioxide (CO2) gas.

Edible Ink
Source Institutions
In this chemistry activity (page 6 of the PDF), learners observe a chemical change. Learners write and reveal a secret message using edible ink.

Bone Basics
Source Institutions
This is an activity (on page 2 of the PDF under Bone Regrowth Activity) about the two main components of bone - collagen and minerals (like calcium) - and how they each contribute to its flexibility a
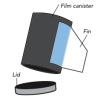
Pop Rockets
Source Institutions
In this activity, learners make film canister rocket ships. A fin pattern is glued onto the outside of the canister, and fuel (water and half an antacid tablet) is mixed inside the canister.

pHun with Cabbage
Source Institutions
In this chemistry activity, learners will test the pH of various foods and household substances using cabbage.

The Effects of Acid Rain
Source Institutions
In this environmental science activity (page 4 of the PDF), learners use vinegar and chalk to observe the effect of acid rain on various building materials and plant life.

Iron in the Environment
Source Institutions
In this chemistry activity (on page 2 of the PDF), learners corrode a penny in a cup with vinegar, salt water, and a source of iron (nails, paper clips, or twist ties).

Fill 'er Up!
Source Institutions
Learners discover that their breath contains carbon dioxide, one of the pollutants found in car exhaust.
