Search Results
Showing results 1 to 20 of 30

Skin Deep
Source Institutions
In this activity, learners explore how to protect their skin while applying pesticides to plants.
Trading Places: Redox Reactions
Source Institutions
Visitors add drops of copper sulfate solution onto a steel nail. They observe the nail change color from silver to brown as the copper plates onto the nail.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

Hot and Cold
Source Institutions
In this chemistry challenge, learners discover that many chemical reactions involve heat loss or gain.

Homemade Hovercraft!
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Hovercraft) is a full inquiry investigation into hovercraft engineering and design optimization.

Wild Sourdough
Source Institutions
In this activity, learners explore chemistry and the microbial world by making their own sourdough starter and bread at home using only flour and water.

Plaster of Paris
Source Institutions
In this activity (page 6 of the PDF), learners will observe both a chemical and a physical change.
Heavy Lifting
Source Institutions
In this activity, learners work in NASA teams to build balloon-powered rockets using identical parts and compete to launch the greatest number of paper clips to "space" (the ceiling).

Kites
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Kites) is a full inquiry investigation into how a kite’s shape affects its performance.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Shrinkers
Source Institutions
In this hands-on activity, learners use heat to shrink samples of polystyrene plastic (#6 recycle code). Learners compare the size and shape of the plastic pieces before and after shrinking.
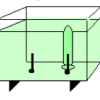
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.
Take Out the Trash
Source Institutions
Learners explore how recyclers take advantage of the different properties of materials, such as magnetism and density, to separate them from a mixture.

Whodunit?
Source Institutions
In this fascinating and fun experiment, learners use chemistry to identify a mystery powder and to solve a "crime," a process similar to that used by real forensic scientists.

Changing Colors
Source Institutions
In this challenge, learners have to figure out in what order to combine five solutions to change the color from clear, to yellow, to blue, and back to clear.

Fruit Juice Mystery
Source Institutions
In this chemistry challenge, learners work to figure out which of four juices are real, and which is just food coloring and sugar.

Dye Detective
Source Institutions
Learners analyze mixtures of dyes using filter paper chromatography. They place spots of the different dyes at the bottom of a piece of filter paper, and hang the paper to touch the surface of water.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.
