Search Results
Showing results 1 to 20 of 27
Trading Places: Redox Reactions
Source Institutions
Visitors add drops of copper sulfate solution onto a steel nail. They observe the nail change color from silver to brown as the copper plates onto the nail.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

Homemade Hovercraft!
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Hovercraft) is a full inquiry investigation into hovercraft engineering and design optimization.
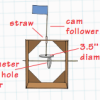
Crank It Up
Source Institutions
In this engineering activity, learners explore simple machines and then build cardboard automata using cams.
First Impressions
Source Institutions
Learners experiment with a commercial photo-sensitive paper (Sunprint® or NaturePrint® paper). They place opaque and clear objects on the paper and expose it to bright light, observing the results.

Fruit Xylophone: Fruit Salad Instrument of the Future!
Source Institutions
This is a perfect summertime lunch activity! Pico Cricket is required (micro controller). First, get a bunch of cut up fruit, line them up, then plug a piece of fruit with a Pico Cricket sensor clip.
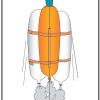
Heavy Lifting
Source Institutions
In this activity, learners work in NASA teams to build balloon-powered rockets using identical parts and compete to launch the greatest number of paper clips to "space" (the ceiling).

Forms of Carbon
Source Institutions
In this activity, educators can demonstrate how the nanoscale arrangement of atoms dramatically impacts a material’s macroscale behavior.

Kites
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Kites) is a full inquiry investigation into how a kite’s shape affects its performance.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Shrinkers
Source Institutions
In this hands-on activity, learners use heat to shrink samples of polystyrene plastic (#6 recycle code). Learners compare the size and shape of the plastic pieces before and after shrinking.

Cooking With the Sun
Source Institutions
In this activity, learners build a simple solar oven out of household materials to melt chocolate and marshmallow between graham crackers--known as s'mores.
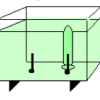
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.

Vocal Visualizer
Source Institutions
With a bit of PVC, a laser, a can/cup, and a small mirror, you can make a device that visualizes you voice or any sound transmitted into the cup/can.
Properties of Metals
Source Institutions
In this activity, learners explore the properties of metals at four stations. The stations include A) Magnetism and Breakfast Cereal; B) Conductivity of Metals; C) Alloys; and D) Metal Plating.

Blimp Jet Challenge
Source Institutions
In this design challenge activity, learners add a jet-propulsion system (i.e. a balloon) to a blimp so it flies straight and far under its own power.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
