Search Results
Showing results 1 to 16 of 16

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.
Shocking Fruit
Source Institutions
In this activity, learners discover how a piece of fruit can act as an electrolyte, conducting electricity between two different metals.
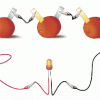
Electricity: Fruit Batteries
Source Institutions
In this activity, learners create a battery from fruit. This activity helps learners explore electricity, electrochemistry, and series circuits as well as the process of scientific inquiry.
Neural Network Signals
Source Institutions
In this activity, learners create an electrical circuit and investigate how some dissolved substances conduct electricity.
Properties of Metals
Source Institutions
In this activity, learners explore the properties of metals at four stations. The stations include A) Magnetism and Breakfast Cereal; B) Conductivity of Metals; C) Alloys; and D) Metal Plating.

Cake by Conduction
Source Institutions
In this demonstration, cook a cake using the heat produced when the cake batter conducts an electric current.

Hand Battery
Source Institutions
In this activity about chemistry and electricity, learners form a battery by placing their hands onto plates of different metals.

Make Your Own Batteries!
Source Institutions
This activity (on page 3 of the PDF under GPS: Body Electricity Activity) is a full inquiry investigation into conductivity.

The Power of Graphene
Source Institutions
This lesson focuses on graphene and its electrical properties and applications.

Exploring Materials: Graphene
Source Institutions
In this activity, learners investigate the properties of graphene and graphite.

Currently Working
Source Institutions
Learners test solutions of water, sugar, salt, and hydrochloric acid for electrical conductivity. They immerse leads from a lighting device (a battery pack connected to an LED) into each solution.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.

Build a Battery
Source Institutions
Learners make a simple battery out of "sandwiches" of aluminum foil, pennies, and a salt water-soaked paper towel.

Fruity Electricity
Source Institutions
In this activity, Frankenstein's lab is running out of electricity! Learners use fruit to help Igor find a temporary source of energy to turn on a light.

Power To Go
Source Institutions
Learners observe an electrochemical cell constructed from a small jar containing zinc and copper strips immersed in separate solutions. The strips are connected to a motor that turns a small fan.

Carrying Charges: Testing for Conductivity
Source Institutions
Learners are challenged to create solutions that conduct electricity and make a buzzer buzz (or an LED light up).
