Search Results
Showing results 21 to 40 of 82
What's So Special about Water: Absorption
Source Institutions
In this activity about water's cohesive and adhesive properties and why water molecules are attracted to each other, learners test if objects repel or absorb water.

Testing Falling Peanut Butter Sandwich Myth
Source Institutions
In this activity related to rotational inertia (page 1 of the PDF under SciGirls Activity: Microgravity), learners will use a bit of scientific experimenting to test if open-faced peanut butter sandwi

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Crystal Gardens
Source Institutions
In this activity, which requires adult supervision, learners get to explore the awesome power of chemistry.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.

Temperature Affects the Solubility of Gases
Source Institutions
In this activity, learners heat and cool carbonated water to find out whether temperature has an effect on how fast the dissolved gas leaves carbonated water.
Wave on Wave
Source Institutions
In this activity, learners use raisins and seltzer water to understand why waves don’t move objects forward. Learners conduct two simple experiments to understand the circular movement of waves.

Twirling Rope Frequency
Source Institutions
In this activity (page 1 of the PDF under SciGirls Activity: Double Dutch), learners will stand twelve feet apart swinging a rope at the slowest tempo possible while someone uses a stopwatch to record

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.

Ziptop Bag Chemistry
Source Institutions
In this chemistry activity, learners perform three chemical reactions in a sealed zip-top bag. Learners will record their observations and classify the changes as chemical or physical.

What Causes Wind?
Source Institutions
In this sunny day experiment, learners measure and compare how quickly light and dark colored materials absorb heat.

What does Color have to do with Cooling?
Source Institutions
In this demonstration/experiment, learners discover that different colors and materials (metals, fabrics, paints) radiate different amounts of energy and therefore, cool at different rates.

Moving Molecules!
Source Institutions
In this activity about molecular diffusion (located on page 2 of the PDF under Nanosilver Activity), learners will make predictions and move molecules of iodine through a seemingly solid plastic sandw
Cooling the Mummy's Tomb
Source Institutions
In this activity, learners conduct an experiment to help Pharaoh design a better insulated tomb.

Tasty Buds
Source Institutions
In this activity (1st activity on the page), learners explore their sense of taste and the structure of the tongue by taste-testing various foods.

In Proportion
Source Institutions
Through this nutrition activity (page 5 of the PDF), learners will understand—and probably be surprised by—how big serving sizes of various foods should be.

Atoms and Matter (K-2)
Source Institutions
In this activity, learners explore atoms as the smallest building blocks of matter. With adult help, learners start by dividing play dough in half, over and over again.

Fingerprints
Source Institutions
In this activity, learners examine their fingerprints and learn that they can be categorized by shape, but each fingerprint is unique.

Think Fast!: Just How Quick Are You?
Source Institutions
This is an activity about reaction times. Just how quickly must an NHL goalie respond to save a shot, and how does your reaction time compare?
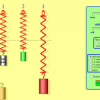
Masses & Springs
Source Institutions
In this online activity, learners use a realistic mass and spring laboratory. They hang masses from springs and adjust the spring stiffness and damping.
