Search Results
Showing results 1 to 20 of 59

Cleaning with Dirt
Source Institutions
Learners build a filter from old soda bottles and dirt. They create polluted water, and pour it through their filter to clean it.
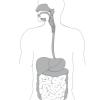
Digestion
Source Institutions
In this food science activity, learners explore digestion and proteins by observing the action of meat tenderizer on luncheon meat.

Modeling Day and Night
Source Institutions
In this activity (on page 1 of the PDF), learners make a "mini-globe" to investigate the causes of day and night on our planet.

Matter on the Move
Source Institutions
Learners observe and conduct experiments demonstrating the different properties of hot and cold materials.

Chromatography Can Separate!
Source Institutions
In this chemistry activity, learners use thin layer chromatography to determine the molecular composition of different markers.

Absorb & Repel
Source Institutions
In this activity, learners investigate how different materials repel or absorb water. Learners use spray bottles to explore how everyday items like sponges, cardboard, feathers, etc.

Digit's Cyber-Dough
Source Institutions
In this fun hands-on activity, learners whip up a batch of cyber-dough (play dough) using math for measurements.

The Scoop on Habitat
Source Institutions
Some aquatic organisms live in open water, while some live in soil at the bottom of a body of water.

M&M's in Different Temperatures
Source Institutions
Learners design their own experiment to investigate whether the temperature of the surrounding water affects the rate at which the colored coating dissolves from an M&M.

Drying It Out
Source Institutions
In this activity, learners investigate and compare the rate of drying in different conditions.
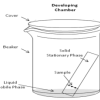
Separating with Chromatography
Source Institutions
In this experiment, learners separate different types of molecules in marker inks (using a technique called "thin layer chromatography").

Comparing the Density of an Object to the Density of Water
Source Institutions
Learners compare the weight of equal volumes of wax, water, and clay. Learners discover that since the wax weighs less than an equal volume of water, it is less dense than water and will float.

Formation of a Precipitate
Source Institutions
Learners create hard water by mixing Epsom salt and water. Then they compare what happens when soap solution is mixed with hard water and regular water.

Defining Density
Source Institutions
In this introductory demonstration and activity, learners are introduced to the concept of density as they explore a rock and a wooden block in water.

Changing the Density of an Object: Changing Shape
Source Institutions
Learners will see that changing the shape of an object, like a clay ball, that is more dense than water, can affect whether the object will sink or float.

Funny Putty
Source Institutions
In this chemistry activity (page 1 of PDF SciGirls Activity: Milk Carton Boat), learners will create a blob of stretchable funny putty out of a water, borax, and glue mixture.

Traveling Through Different Liquids
Source Institutions
Learners observe and record what happens when they manipulate bottles containing a liquid (water or corn syrup) and one or more objects (screw, nail, paper clip).

Kool Colors
Source Institutions
Learners investigate how temperature affects the rate of chemical reactions by observing how steel wool reacts with various types of Kool-Aid solutions at different temperatures.

Spaghetti Bridge
Source Institutions
In this engineering activity, learners investigate the differences between the strength of bridges made from flat and round building materials.

Evaporation
Source Institutions
This three-part activity consists of an activity that groups of learners develop themselves, a given procedure, and an optional demonstration.
