Search Results
Showing results 1 to 11 of 11

Glow Fast, Glow Slow: Alter the Rate of a Reaction!
Source Institutions
Learners investigate one factor affecting reaction rates: temperature. In a darkened room, two identical lightsticks are placed in water -- one in hot water and one in cold water.

Rate of Solution Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the factors that increase the rate of dissolution for a solid.
Floating Candles
Source Institutions
In this chemistry activity, learners observe a combustion reaction and deduce the components necessary for the reaction to occur.

Half Full or Half Empty
Source Institutions
In this activity (12th activity on the page), learners conduct an experiment to demonstrate how muscles are constantly feeding information to the brain about what they are doing.

Potato Power
Source Institutions
Learners combine hydrogen peroxide with three different forms of potato: raw chunks, ground chunks, and boiled chunks.

Does Air Weigh Anything?
Source Institutions
The demonstration/experiment provides quick proof that air has mass.

Auditory Acuity
Source Institutions
This activity (8th activity on the page) tests learners' ability to identify things using only the sense of hearing.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.

Laser Jello
Source Institutions
In this activity, learners use gelatin as a lens to investigate the properties of laser light.

Double Replacement Micro-Reactions
Source Institutions
In this chemistry activity, learners use common chemicals to examine reactions that occur between two aqueous solutions.
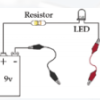
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.
