Search Results
Showing results 1 to 20 of 82

Say Cheese!
Source Institutions
Create a chemical reaction that makes cheese! This hands-on activity demonstrates that molecules and atoms are tiny particles that make up everything around us.

Recrystallization Test
Source Institutions
In this activity, learners recrystallize substances from solutions and make observations about the resulting crystals. This test can help further identify the unknown.

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Density: Make a golf ball float!
Source Institutions
In this activity (on page 2 of the PDF), the learner places a golf ball between salt water and colored fresh water. The golf ball is not as dense as the saltwater.

Egg Drop
Source Institutions
In this activity, kids make and play with Ooze before testing the material in an egg drop!
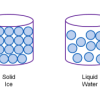
Atoms and Matter (3-6)
Source Institutions
In this activity, learners build models of atoms and molecules, then consider their role in different phases of matter, density, and mixtures and solutions.

Polymers are Chains (K-2)
Source Institutions
In this activity, learners make a paper model of a polymer, then make Silly Putty, an actual polymer.

Cool Hot Rod
Source Institutions
If you have access to a copper metal tube, this activity does a great job demonstrating what happens to matter when it's heated or cooled. This activity requires some lab equipment.

Rubber Band Thermodynamics
Source Institutions
In this demonstration, learners explore the thermal properties of rubber. Learners investigate whether a rubber band contracts or expands when heated.
Twirling in the Breeze
Source Institutions
In this engineering activity, learners build a device (an anemometer) to measure how fast the wind is blowing.

Submarine: Soda Cup Lander
Source Institutions
In this activity (on page 2), learners create a submarine using a plastic cup. This is a fun way to learn about buoyancy and density.
Deep Sea Diver
Source Institutions
In this ocean engineering activity, learners explore buoyancy and water displacement. Then, learners design models of deep sea divers that are neutrally buoyant.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.

Toy Chemistry
Source Institutions
In this playful, goopy activity, learners mix two liquids to create a solid (that sometimes acts like a liquid ), using basic household materials such as borax and glue.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

Scream for Ice Cream
Source Institutions
Don't scream for ice cream -- make it with milk, sugar, flavoring and some 'salt-water' ice. Discover the chemistry of ice cream by creating your own.

ZOOM Glue
Source Institutions
In this activity, learners mix milk, vinegar, baking soda, and water to create sticky glue. Use this activity to explain how engineers develop and evaluate new materials and products.

Supercooled Water Drops
Source Institutions
In this activity, learners touch supercooled water drops with an ice crystal and trigger the water drops to freeze instantly.

Bubble Trouble
Source Institutions
In this activity on page 15 of the PDF, learners measure the amount of bubbles that they make using a detergent.

What Counts in Bounce
Source Institutions
In this activity learners compare the bounciness of warm and cold racquetballs to see if temperature makes a difference in how well they bounce.
