Search Results
Showing results 1 to 20 of 25

Nano Ice Cream
Source Institutions
In this activity/demo, learners discover how liquid nitrogen cools a creamy mixture at such a rapid rate that it precipitates super fine grained (nano) ice cream.

Illuminations on Rates of Reactions
Source Institutions
In this activity, learners investigate the speed of chemical reactions with light sticks. Learners discover that reactions can be sped up or slowed down due to temperature changes.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Phase Changes
Source Institutions
Learners observe a sealed test tube containing a small amount of solid stearic acid.

Chemistry in the Kitchen
Source Institutions
In this kitchen chemistry activity, learners explore the chemistry of crystals by making sugar crystals, consider a common chemical reaction type responsible for the rising of muffins and cake in the

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.

Full of Hot Air: Hot Air Balloon Building
Source Institutions
In this activity, learners create a model of a hot air balloon using tissue paper and a hairdryer. Educators can use this activity to introduce learners to density and its role in why things float.

Tempest in a Teacup
Source Institutions
In this hands-on activity, learners determine the types of chemical reactions achieved when combining different household products.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.

That's the Way the Ball Bounces: Level 2
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Biochemistry Happens Inside of You!
Source Institutions
In this four-part activity, learners explore how the body works and the chemistry that happens inside living things.

We all Scream for Ice Cream
Source Institutions
In this activity, learners observe how salinity affects the freezing point of water by making and enjoying ice cream.

Indicating Electrolysis
Source Institutions
In this activity, learners build a simple electrolysis device. Then learners use an indicating solution to visualize hydrogen and oxygen molecules in water.

Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.
Sodium Acetate Hand Warmers
Source Institutions
In this activity, sodium acetate hand warmers are used to introduce learners to supersaturated solutions, crystallization, and exothermic reactions.
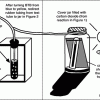
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.

Edible Ink
Source Institutions
In this chemistry activity (page 6 of the PDF), learners observe a chemical change. Learners write and reveal a secret message using edible ink.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
