Search Results
Showing results 1 to 20 of 34

Clear Slime Polymer
Source Institutions
In this chemistry activity, learners use guar gum to make slime. Use this activity to introduce learners to polymers, viscosity, and colloids.

Gravity Fail
Source Institutions
In this activity, learners try pouring water out of a regular cup and a miniature cup. It’s harder than it sounds! Learners discover that different forces dominate at different size scales.

Introduction to the New Chain Gang
Source Institutions
In this activity, learners use pop-beads to understand the characteristics and properties of polymer chains.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.

Oil Slick
Source Institutions
Can you think of ways to collect and dispose of spilled oil without causing further harm to the environment?

Chemistry Cake
Source Institutions
In this exciting and tasty chemistry activity which requires adult supervision, learners explore how chemistry affects a simple everyday activity like cooking.

Chemistry in the Kitchen
Source Institutions
In this kitchen chemistry activity, learners explore the chemistry of crystals by making sugar crystals, consider a common chemical reaction type responsible for the rising of muffins and cake in the

That's the Way the Ball Bounces: Level 1
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, size, and bounce height.

Chemistry Is Colorful
Source Institutions
In "Chemistry is Colorful" learners explore different materials through paper chromatography.
Choose Your Ooze
Source Institutions
During this activity, learners will make different versions of "ooze" using varied proportions of detergent and glue.
Stability of Egg White Foams
Source Institutions
In this chemistry meets cooking activity, learners compare the stability of egg white foams with various additives.
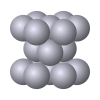
Crystal Packin' Mama
Source Institutions
In this activity, learners investigate the basic crystal structures that metal atoms form.

That's the Way the Ball Bounces: Level 2
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Liquid Lava Layers
Source Institutions
In this activity, learners explore the concepts of density and basic chemical reactions as they create a homemade lava lamp effect using water, oil, food coloring, and Alka-Seltzer tablets.

Crunch and Munch Lab
Source Institutions
In this activity, learners use three types of cheesy snacks--cheese balls, cheese puffs, and Cheetos--to learn about polymers.
Glitter Slime
Source Institutions
In this activity on page 8 of the PDF, learners make a slimy substance very similar to mucus, and sprinkle it with glitter to imitate the way that allergens are trapped.

Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.

Exploring the Nanoworld with LEGO Bricks: Structure-Property Relationships at the Nanoscale
Source Institutions
In this activity (pages 32-41), learners learn how the atomic and molecular arrangement of matter are related to physical properties.
