Search Results
Showing results 1 to 20 of 28

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.

It's a Gas, Man
Source Institutions
In this activity, learners discover if carbon dioxide has an effect on temperature.
Sea State: Forecast Conditions at Sea
Source Institutions
In this oceanography and data collection activity, learners cast real time sea state conditions using buoys from NOAA's National Data Buoy Center.

Rate of Solution Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the factors that increase the rate of dissolution for a solid.
Understanding Albedo
Source Institutions
In this activity related to climate change, learners examine albedo and the ice albedo feedback effect as it relates to snow, ice, and the likely results of reduced snow and ice cover on global temper

Shrinky Dinks
Source Institutions
Heat makes some materials expand, and it makes others shrink.
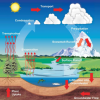
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.
Floating Candles
Source Institutions
In this chemistry activity, learners observe a combustion reaction and deduce the components necessary for the reaction to occur.

Pie-Pan Convection
Source Institutions
It's difficult to see convection currents in any liquid that's undergoing a temperature change, but in this Exploratorium Science Snack, you can see the currents with the help of food coloring.

From Gas to Liquid to Solid
Source Institutions
What causes frost to form on the outside of a cold container? In this activity, learners discover that liquid water can change states and freeze to become ice.

The Carbon Cycle and its Role in Climate Change: Activity 1
Source Institutions
In this activity (on page 1), learners role play as atoms to explore how atoms can be rearranged to make different materials.

Melting Ice
Source Institutions
In this activity, learners explore density, convection, stratification, and, by inference, the melting of icebergs. Learners make hypotheses, test their hypotheses, and explain their observations.

Sinking Water
Source Institutions
In this experiment, learners float colored ice cubes in hot and cold water.

Convection Demonstration
Source Institutions
In this quick activity (located on page 2 of the PDF under GPS: Balloon Fiesta Activity), learners will see the effects of convection and understand what makes hot air balloons rise.
Physical Change
Source Institutions
In this activity, learners use heat to separate zinc and copper in a penny. This experiment demonstrates physical properties and how physical change (phase change) can be used to separate matter.

That Sinking Feeling
Source Institutions
In this quick activity, learners observe how salinity and temperature affect the density of water, to better understand the Great Ocean Conveyor.

Inverted Bottles
Source Institutions
In this activity, learners investigate convection by using food coloring and water of different temperatures.

Sizing Up Temperature
Source Institutions
In this activity, learners explore Charles' Law in a syringe.
Let's Go Ice Fishing
Source Institutions
In this activity, learners are challenged to lift a floating ice cube out of a glass of water using just one end of a piece of string.

Super Shrinkers
Source Institutions
In this activity on page 14 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners turn plain pieces of recycled plastic into shrunken works of art.
