Search Results
Showing results 1 to 20 of 22

Hot Stuff!: Investigation #4
Learners test two jars containing soil, one covered and one open, for changes in temperature. After placing the jars in the Sun, learners discover that the covered jar cools down more slowly.

Dripping Wet or Dry as a Bone?
Learners investigate the concept of humidity by using a dry and wet sponge as a model. They determine a model for 100% humidity, a sponge saturated with water.

Hot Stuff!: Investigation #1
Learners test two jars, one containing plain air and one containing carbon dioxide gas, to see their reactions to temperature changes.

Pinhole Viewer
Source Institutions
In this activity, learners discuss and investigate how cameras, telescopes, and their own eyes use light in similar ways.
Sea State: Forecast Conditions at Sea
Source Institutions
In this oceanography and data collection activity, learners cast real time sea state conditions using buoys from NOAA's National Data Buoy Center.

Weighty Questions
Source Institutions
In this activity about humans and space travel (page 1 of PDF), learners compare and contrast the behavior of a water-filled plastic bag, both outside and inside of a container of water.

A Merry-Go-Round for Dirty Air
Learners build a model of a pollution control device--a cyclone. A cyclone works by whirling the polluted air in a circle and accumulating particles on the edges of the container.

Mint Your Own Coin
Source Institutions
Coins are everyday objects which tell a lot about the people who use them.
Turning the Air Upside Down: Convection Current Model
Learners see convection currents in action in this highly visual demonstration. Sealed bags of colored hot or cold water are immersed in tanks of water.
Hazardous Chemicals in Your Neighborhood
Source Institutions
In this environmental science lesson, learners will examine hazardous chemicals and their effects on human health and the environment.

Hot Stuff!: Carbon Dioxide Extinguishes a Flame
In this demonstration, learners observe vinegar and baking soda creating carbon dioxide (CO2) in a bottle. The gas is poured out of a bottle onto a candle flame, putting out the candle.

Hot Stuff!: Investigation #2
Learners test two jars containing hot water, one covered with plastic and one open, for changes in temperature.
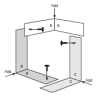
Building a Magic Carpet
Source Institutions
In this activity (page 89 of the PDF), learners compare and contrast pitch and roll motions by using a Magic Carpet maze similar to one that was used for Neurolab investigations about microgravity.

Hot Stuff!: Creating and Testing for Carbon Dioxide
In this demonstration, learners observe vinegar and baking soda reacting to form carbon dioxide (CO2) gas.
Building a 3-D Space Maze: Escher Staircase
Source Institutions
In this activity (page 95 of the PDF), learners create Escher Staircase models similar to those that were used by Neurolab's Spatial Orientation Team to investigate the processing of information about
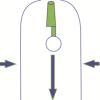
Cartesian Diver
Source Institutions
In this quick activity (page 1 of the PDF under SciGirls Activity: California Fish), learners will build a simple Cartesian Diver in an empty 2-liter bottle.

Turning the Air Upside Down: Warm Air is Less Dense than Cool Air
Learners cover a bottle with a balloon. When they immerse the bottle in warm water, the balloon inflates. When they immerse the bottle in a bowl of ice, the balloon deflates.

Dancing Cereal
Source Institutions
In this quick activity (on page 2 of the PDF under GPS: Body Electricity Activity), learners will observe how dry breakfast cereal appears to dance when it gets close to a balloon charged with static

Hot Stuff!: Investigation #3
Learners test two jars of ice water, one covered and one open, for changes in temperature. After placing the jars in the sun, learners discover that the covered jar cools down more slowly.

Oil Spot Photometer
Source Institutions
In this math activity related to light, learners assemble a photometer and use it to estimate the power output of the Sun.
